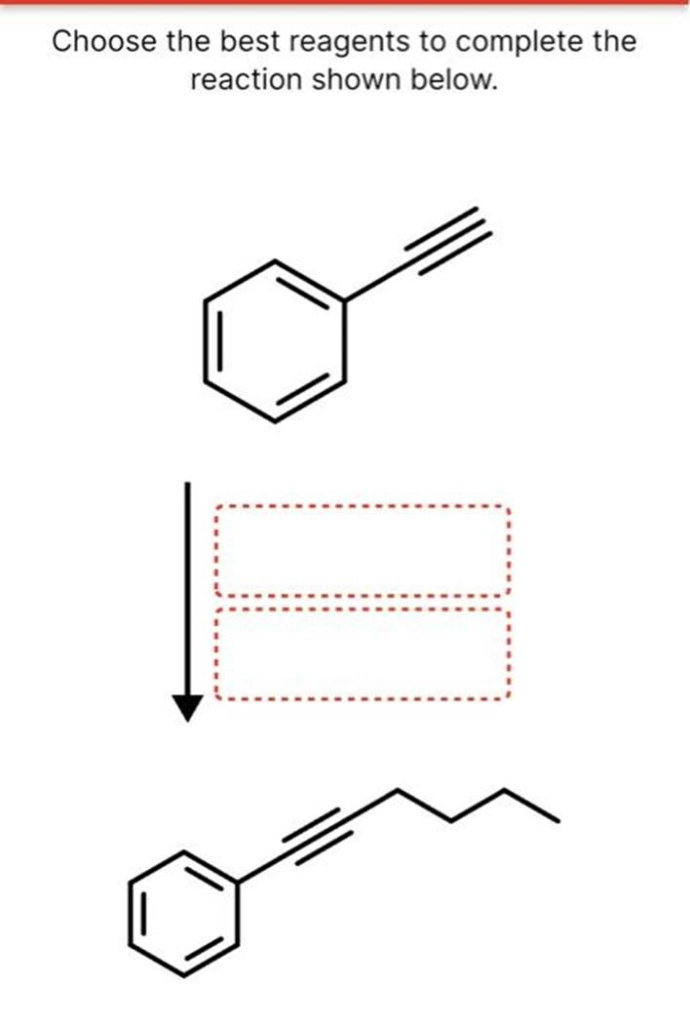
Choose the best reagents to complete the reaction shown below. Select to Draw Select to Draw H/P: HCI (excess) HBr (1 equiv) H/Lindlar’s catalyst Select to Draw Select to Draw Choose the best reagents to complete the reaction shown. Br Br Predict the major product for this reaction. Ignore inorganic byproducts. 1. BH3-THE 2. H2O2, NaOH Select to Draw Draw the major product for this reaction. Ignore inorganic byproducts. 1. Hg(OAC)2, H20 2. H2SO4 Select to Draw
The Correct Answer and Explanation is :
Here’s a list of answers to each clue, along with explanations:
- Symbol Au
Answer: Gold
Gold has the symbol Au, derived from its Latin name aurum. It is a highly valued metal known for its use in jewelry and currency. - The most reactive nonmetal
Answer: Fluorine
Fluorine (F) is the most reactive nonmetal on the periodic table. It reacts violently with many substances, including metals and organic compounds. - Atomic number of 12
Answer: Magnesium
Magnesium (Mg) has an atomic number of 12. It is a shiny gray metal that is essential for many biological processes. - An alkali metal in period 4
Answer: Potassium
Potassium (K) is an alkali metal in period 4. It is highly reactive, especially with water. - The most abundant element in air. Hint: it has an atomic number of 7
Answer: Nitrogen
Nitrogen (N) has an atomic number of 7 and makes up about 78% of the Earth’s atmosphere. - A metal with 3 valence electrons, used in cans
Answer: Aluminum
Aluminum (Al) has 3 valence electrons and is commonly used to make cans due to its lightweight and corrosion-resistant properties. - A period 3 element that will not react naturally
Answer: Argon
Argon (Ar) is a noble gas in period 3. As a noble gas, it is chemically inert and does not react under normal conditions. - Has 5 valence electrons and is in period 3
Answer: Phosphorus
Phosphorus (P) has 5 valence electrons and is located in period 3. It is crucial in biological processes like the formation of DNA. - The most reactive metal
Answer: Francium
Francium (Fr) is the most reactive metal. It is an alkali metal and highly unstable, rarely found in nature. - A period 1 element used to make balloons float
Answer: Hydrogen
Hydrogen (H) is a period 1 element, and its low density makes it suitable for filling balloons that float. - The only nonmetal in group 1
Answer: Hydrogen
Hydrogen (H) is the only nonmetal in group 1 of the periodic table. Despite being in the alkali metal group, it behaves differently from metals. - Responsible for the smell of rotten eggs. Hint: it has 16 protons
Answer: Sulfur
Sulfur (S) has 16 protons and is responsible for the characteristic odor of rotten eggs, often found in compounds like hydrogen sulfide. - Has an atomic number that doubles silicon’s
Answer: Germanium
Germanium (Ge) has an atomic number of 32, which is double that of silicon (Si), which has an atomic number of 14. - The first element in the group of rare earth metals
Answer: Lanthanum
Lanthanum (La) is the first element in the rare earth metal group, located in the f-block of the periodic table. - A group 12 metal that was once used in thermometers, but is now known to be poisonous
Answer: Mercury
Mercury (Hg) is a group 12 metal that was once used in thermometers. It is now known to be highly toxic. - Has 3 valence electrons and 4 energy levels
Answer: Gallium
Gallium (Ga) has 3 valence electrons and is in period 4, meaning it has 4 energy levels. - The only metalloid in period 3
Answer: Silicon
Silicon (Si) is the only metalloid in period 3. It has properties of both metals and nonmetals, making it useful in electronics. - The only metalloid with 3 valence electrons
Answer: Boron
Boron (B) is the only metalloid with 3 valence electrons. It is essential in the manufacture of glass and ceramics. - The alkali metal that makes up table salt
Answer: Sodium
Sodium (Na) is an alkali metal that combines with chlorine to form sodium chloride (NaCl), the compound we know as table salt. - Its most common isotope has 5 neutrons
Answer: Boron
Boron (B) has a common isotope with 5 neutrons. Its atomic number is 5, meaning it has 5 protons, and the most common isotope has 5 neutrons.
These clues provide a great overview of key elements on the periodic table. Each answer is based on the element’s unique properties, atomic number, or place in the table.