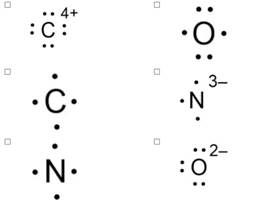
1.Select the atoms or ions drawn with valid Lewis dot structures.
2.a) Draw the Lewis structure of acetone, (CH3)2CO
Step 1: Identify the number of valence electrons.
Step 2: Identify the central atom.
Step 3: Establish connectivity of single bonds.
Step 4: Identify the number of electrons unaccounted for.
Step 5: Complete the structure.
b) Identify the central atom in (CH3)2CO, which is it out of the following?
carbon
oxygen
hydrogen
The Correct Answer and Explanation is :
1. Validity of Lewis Dot Structures:
The image you’ve provided contains several atoms and ions. To determine which have valid Lewis dot structures, we need to consider the following:
- Valence Electrons: Each atom’s valence electrons are represented as dots around the element symbol.
- Octet Rule: Atoms (except hydrogen) tend to form bonds to achieve a full valence shell of eight electrons.
- Formal Charges: The distribution of electrons should minimize formal charges, leading to a more stable structure.
Without specific details about each atom or ion in the image, I can’t provide a definitive assessment. However, atoms like noble gases (e.g., Ne, Ar) naturally have complete valence shells and thus have stable Lewis structures. Ions such as Na⁺ or Cl⁻ also have valid Lewis structures, with Na⁺ having no dots (indicating a full valence shell) and Cl⁻ having eight dots.
2. Drawing the Lewis Structure of Acetone ((CH₃)₂CO):
a) Steps to Draw the Lewis Structure:
- Identify the Number of Valence Electrons:
- Carbon (C): 4 valence electrons × 3 atoms = 12 electrons
- Hydrogen (H): 1 valence electron × 6 atoms = 6 electrons
- Oxygen (O): 6 valence electrons × 1 atom = 6 electrons
- Total valence electrons = 12 + 6 + 6 = 24 electrons
- Identify the Central Atom:
- Oxygen (O) is the central atom, as it is double-bonded to one carbon and single-bonded to another.
- Establish Connectivity of Single Bonds:
- Connect the two methyl groups (CH₃) to the central carbonyl group (C=O).
- Identify the Number of Electrons Unaccounted For:
- Each single bond uses 2 electrons.
- There are 3 single bonds (2 C-H and 1 C-C), using 6 electrons.
- The double bond (C=O) uses 4 electrons.
- Total electrons used = 6 + 4 = 10 electrons.
- Electrons remaining = 24 – 10 = 14 electrons.
- Complete the Structure:
- Distribute the remaining electrons as lone pairs on the oxygen atom and the carbon atoms to satisfy the octet rule.
b) Identifying the Central Atom in Acetone ((CH₃)₂CO):
The central atom in acetone is carbon. It is double-bonded to oxygen and single-bonded to two other carbon atoms, forming a ketone functional group. This central carbon atom is sp² hybridized, leading to a trigonal planar geometry around it. The oxygen atom is double-bonded to this central carbon, and each of the two methyl groups (CH₃) is single-bonded to the central carbon. This arrangement results in a molecule with a trigonal planar structure around the central carbon atom.
Understanding the central atom’s role is crucial in determining the molecule’s geometry and reactivity. In acetone, the central carbon atom’s sp² hybridization and the presence of the carbonyl group (C=O) contribute to its polarity and solvent properties. Acetone’s polarity allows it to dissolve a wide range of substances, making it an effective solvent in various industrial and laboratory applications.