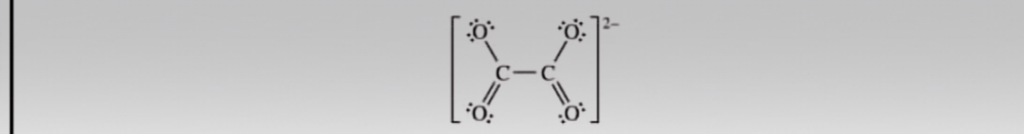
A Lewis electron-dot diagram of the oxalate ion, C_{2}O_{4} ^ 2- is shown. (a) Identify the hybridization of the valence orbitals of either carbon atom in the oxalate ion. (b) Silver oxalate, Ag_{2}C_{2}O_{4}(s) is slightly soluble in water. The value of K sp for Ag_{2}C_{2}*O_{4} is 5.4 * 10 ^ – 12 (i) Write the expression for the solubility-product constant, K sp for Ag_{2}C_{2}O_{4} (ii) Calculate the molar solubility of Ag_{2}C_{2}O_{4} in neutral distilled water.
The Correct Answer and Explanation is :
(a) Hybridization of Carbon in Oxalate Ion (C₂O₄²⁻):
In the oxalate ion (C₂O₄²⁻), each carbon atom is bonded to two oxygen atoms through single bonds and one oxygen atom through a double bond. This leads to the formation of a planar structure with resonance between the two possible structures (where the double bond shifts between the two carbon-oxygen bonds).
Each carbon atom in the oxalate ion forms three sigma bonds: two with oxygen atoms and one with the other carbon atom. The carbon-carbon bond is a sigma bond, and the carbon-oxygen bonds include a sigma bond and a pi bond (for the double-bonded oxygen). This configuration implies that the carbon atoms are involved in three bonding regions, suggesting sp² hybridization for each carbon atom.
(b) Silver Oxalate Solubility:
(i) Expression for the Solubility-Product Constant (Ksp) of Ag₂C₂O₄:
The dissolution of silver oxalate (Ag₂C₂O₄) in water can be represented by the following equilibrium equation:
[
\text{Ag}_2\text{C}_2\text{O}_4 (s) \rightleftharpoons 2 \text{Ag}^+ (aq) + \text{C}_2\text{O}_4^{2-} (aq)
]
The solubility product constant expression is:
[
K_{sp} = [\text{Ag}^+]^2 [\text{C}_2\text{O}_4^{2-}]
]
where:
- ([\text{Ag}^+]) is the concentration of silver ions,
- ([\text{C}_2\text{O}_4^{2-}]) is the concentration of oxalate ions.
(ii) Calculation of Molar Solubility:
Let the molar solubility of Ag₂C₂O₄ in water be represented by (s). According to the dissolution reaction:
- For every 1 mole of Ag₂C₂O₄ that dissolves, 2 moles of Ag⁺ are produced, and 1 mole of C₂O₄²⁻ is produced.
Thus, at equilibrium:
- The concentration of Ag⁺ will be (2s),
- The concentration of C₂O₄²⁻ will be (s).
Substituting these concentrations into the Ksp expression:
[
K_{sp} = (2s)^2 (s) = 4s^3
]
Given that the Ksp value is (5.4 \times 10^{-12}), we can solve for (s):
[
4s^3 = 5.4 \times 10^{-12}
]
[
s^3 = \frac{5.4 \times 10^{-12}}{4} = 1.35 \times 10^{-12}
]
[
s = \sqrt[3]{1.35 \times 10^{-12}} \approx 1.1 \times 10^{-4} \, \text{M}
]
Thus, the molar solubility of Ag₂C₂O₄ in neutral distilled water is approximately (1.1 \times 10^{-4} \, \text{M}).
Explanation:
- Hybridization of Carbon: The carbon atoms in oxalate ion are sp² hybridized because they are involved in three regions of bonding (two sigma bonds with oxygen and one with the other carbon atom), and the structure of oxalate is planar due to resonance.
- Solubility of Silver Oxalate: The solubility product (Ksp) provides a way to quantify how much of a slightly soluble salt (like Ag₂C₂O₄) dissolves in water. The concentration of ions at equilibrium is calculated based on the dissociation of the compound into its ions. By solving for the molar solubility, we find the concentration of silver oxalate that will dissolve in water to be approximately (1.1 \times 10^{-4} \, \text{M}).