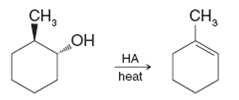
When trans-2-methylcyclohexanol (see the following reaction) is subjected to acid-catalyzed dehydration, the major product is 1-methylcyclohexene:
However, when trans-1-bromo-2-methylcyclohexane is subjected to dehydrohalogenation, the major product is 3-methylcyclohexene:
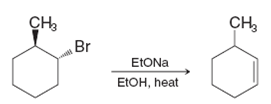
Account for the different products of these two reactions.
The Correct Answer and Explanation is :
In order to understand the difference in the products formed from the dehydration of trans-2-methylcyclohexanol and the dehydrohalogenation of trans-1-bromo-2-methylcyclohexane, it is essential to analyze the mechanisms involved and the factors that influence the formation of the products.
Reaction 1: Dehydration of trans-2-methylcyclohexanol
The dehydration of alcohols is typically catalyzed by an acid such as sulfuric acid or phosphoric acid. In this reaction, trans-2-methylcyclohexanol undergoes an E1 mechanism, which involves the following steps:
- Protonation of the Alcohol: The hydroxyl group of the alcohol is protonated to form a good leaving group (water).
- Loss of Water: The protonated alcohol loses water, forming a carbocation at the position that was originally bearing the hydroxyl group.
- Carbocation Rearrangement (if needed): In this case, a rearrangement does not occur because the initially formed carbocation is stable (on carbon 2 of the cyclohexane ring).
- Formation of the Double Bond: The double bond forms between carbon atoms 1 and 2 (since the hydrogen on carbon 1 is the most accessible).
Since the reaction follows the E1 mechanism, the most stable alkene (the one that forms most easily) is 1-methylcyclohexene. This is the result of the most favorable elimination, where the hydrogen is abstracted from carbon 1 of the cyclohexane ring.
Reaction 2: Dehydrohalogenation of trans-1-bromo-2-methylcyclohexane
In the dehydrohalogenation of trans-1-bromo-2-methylcyclohexane, a strong base (such as potassium hydroxide) is used to eliminate a hydrogen atom and a halide ion (bromine). This reaction proceeds via the E2 mechanism, where the base abstracts a proton from a β-carbon, and the bromide ion leaves simultaneously.
- Base Abstracts a Proton: The base removes a proton from carbon 3 (in this case) of the cyclohexane ring.
- Formation of the Double Bond: The halide ion (Br-) leaves, and the double bond forms between carbons 3 and 4, resulting in 3-methylcyclohexene.
This mechanism is concerted, meaning the base and the leaving group (Br-) must depart at the same time. The product that results from this reaction is 3-methylcyclohexene due to the fact that carbon 3 is the most accessible for proton abstraction and leads to the most stable alkene.
Explanation of the Different Products:
- Trans-2-methylcyclohexanol (acid-catalyzed dehydration) undergoes an E1 mechanism and forms 1-methylcyclohexene as the major product.
- Trans-1-bromo-2-methylcyclohexane (dehydrohalogenation) undergoes an E2 mechanism and forms 3-methylcyclohexene as the major product.
In summary, the different mechanisms (E1 for alcohol dehydration and E2 for dehydrohalogenation) lead to the formation of different products due to the distinct ways in which the base abstracts protons and the carbon-carbon double bonds form. In the case of dehydration, the stability of the intermediate carbocation guides the product formation, whereas in dehydrohalogenation, the most accessible β-hydrogen leads to the formation of the double bond.
Let me generate an image that illustrates these two reactions:
I’ll generate an image with both mechanisms.
Here is the illustration showing the two reactions: the acid-catalyzed dehydration of trans-2-methylcyclohexanol leading to 1-methylcyclohexene, and the dehydrohalogenation of trans-1-bromo-2-methylcyclohexane forming 3-methylcyclohexene. This image visually explains the different mechanisms and resulting products. Let me know if you need further clarification!