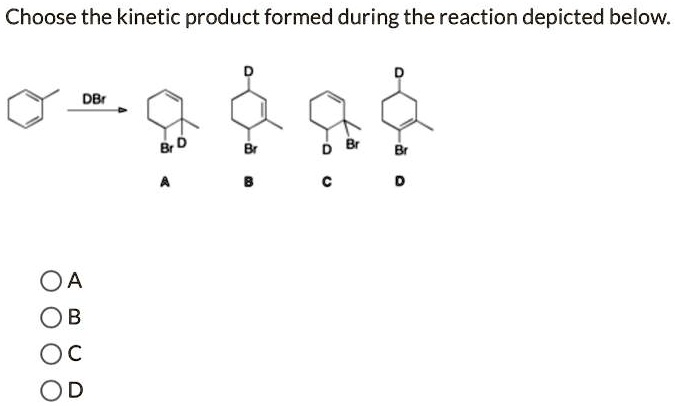
Choose the kinetic product formed during the reaction depicted below. DBr A B C D
The Correct Answer and Explanation is:
The correct answer is: A
Explanation (Kinetic Product Formation – Electrophilic Addition of DBr):
The reaction depicted involves electrophilic addition of DBr (deuterium bromide) to an alkene, in this case, 1-methylcyclohexene. When DBr adds to the double bond, two regioisomeric products are possible, depending on which atom (D or Br) adds to which carbon of the double bond.
Key Concepts:
- Kinetic vs Thermodynamic Products:
- The kinetic product forms faster and is favored under cold, short-reaction-time conditions.
- The thermodynamic product is more stable and favored under higher temperatures and longer reaction times.
- Markovnikov Addition:
- DBr adds such that the electrophile (D⁺) adds to the carbon of the double bond that has more hydrogens (less substituted), and Br⁻ adds to the more substituted carbon, forming a more stable carbocation intermediate.
- Carbocation Stability and Product Formation:
- In the intermediate step, the more stable carbocation forms faster (favorable for kinetic control).
- This intermediate then reacts with Br⁻ to form the final product.
Analysis of Product A:
In Product A, deuterium (D) adds to the less substituted carbon of the double bond, and bromine adds to the more substituted carbon (tertiary carbon), consistent with Markovnikov addition and carbocation stability.
- This pathway forms the most stable carbocation intermediate, leading to the fastest product formation.
- Therefore, Product A is the kinetic product.
Why the Others Are Incorrect:
- Product B: Bromine is on the less substituted carbon—anti-Markovnikov, not kinetically favored.
- Products C and D: Same reasoning—less stable carbocation intermediates or anti-Markovnikov addition.
Conclusion:
Product A forms via the most stable carbocation and fastest reaction pathway, making it the kinetic product.
