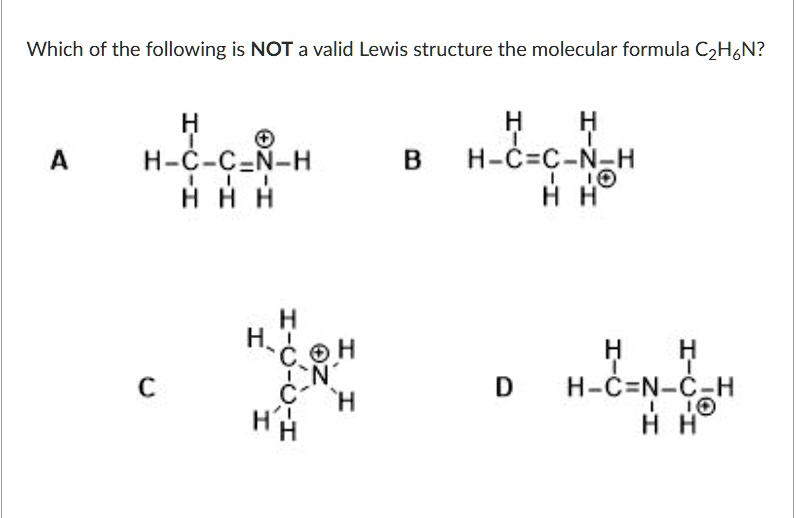
Which of the following is NOT a valid Lewis structure for the molecular formula C2H6N? A: H – C – C = N – H (with H atoms and a positive charge on C) B: H – C = C – N – H (with H atoms and a negative charge on N) C: H2 – C – (C with positive charge) – N – H2 (with H atoms) D: H – C – N = C – H (with H atoms and two charges)
The Correct Answer and Explanation is:
The correct answer is: A
Explanation:
To determine which structure is NOT a valid Lewis structure for the molecular formula C₂H₆N, we need to evaluate each structure based on:
- Octet rule for atoms (especially carbon and nitrogen),
- Correct number of valence electrons,
- Formal charges, and
- Total atom count: 2 carbon (C), 6 hydrogen (H), and 1 nitrogen (N).
Option A:
Structure shows a carbon atom forming five bonds — one single bond with hydrogen, two single bonds with adjacent carbon and hydrogen, and a double bond with nitrogen while also holding a positive formal charge.
- Problem: Carbon cannot have five bonds (10 electrons), as it violates the octet rule.
- Conclusion: ❌ Invalid Lewis structure
Option B:
- The molecule has two carbon atoms, and nitrogen is attached to one carbon and a hydrogen, carrying a negative charge.
- All atoms obey the octet rule, and the total number of atoms matches C₂H₆N.
- Formal charges are plausible.
- Conclusion: ✅ Valid
Option C:
- The central carbon has a positive charge, nitrogen is neutral.
- Atom counts and bonding obey valence and octet rules.
- All atoms appear correctly bonded with correct charges.
- Conclusion: ✅ Valid
Option D:
- Carbon and nitrogen are correctly arranged.
- The nitrogen forms three bonds and a lone pair, carbon maintains four bonds, and the formal charges are plausible.
- Atom count and electron rules are satisfied.
- Conclusion: ✅ Valid
Final Answer: ✅ A is NOT a valid Lewis structure because it violates the octet rule for carbon, making it chemically implausible despite matching the molecular formula C₂H₆N.
