A 20 g mixture containing 95% (by weight) of A and 5% of B is recrystallized in toluene (bp: 110*C). Solubilities in g/100 mL are given below: What amount of solvent is needed to obtain pure A and how much pure A will be recovered ? Show all calculations and explain your answers. Hint: the goal of a recrystallization is to keep ALL of the impurity dissolved at the low temperature: Knowing that; how much solvent is needed for this example? Temperature Solubility A 208 110′ 1.5 g 10.0 g (g/100 mL) 8.0 g 0.5 & Question 4: Name the seven steps required for recrystallization of organic compounds?

The Correct Answer and Explanation is:
Given:
- Total mixture: 20 g
- A = 95% → 0.95 × 20 = 19 g
- B = 5% → 0.05 × 20 = 1 g
Goal: Recrystallize A and keep all of impurity B dissolved at low temperature (20°C).
Step-by-step Calculation:
Step 1: Use solubility of B at 20°C to calculate minimum solvent volume needed to keep all of B dissolved
- Solubility of B at 20°C = 0.5 g/100 mL
- We need to dissolve 1.0 g of B, so:
1.0 g0.5 g/100 mL=200 mL of toluene\frac{1.0 \, g}{0.5 \, g/100 \, mL} = 200 \, \text{mL of toluene}0.5g/100mL1.0g=200mL of toluene
👉 Minimum 200 mL of solvent needed to keep all of B dissolved at 20°C.
Step 2: How much A remains dissolved at 20°C in 200 mL?
- Solubility of A at 20°C = 1.5 g/100 mL
- In 200 mL:
1.5 g100 mL×200 mL=3.0 g\frac{1.5 \, g}{100 \, mL} \times 200 \, mL = 3.0 \, g100mL1.5g×200mL=3.0g
👉 3 g of A will remain dissolved at 20°C and be lost.
Step 3: How much A is recovered?
- Initial A = 19 g
- Dissolved/lost A = 3 g
Recovered A=19 g−3 g=16 g\text{Recovered A} = 19 \, g – 3 \, g = \boxed{16 \, g}Recovered A=19g−3g=16g
✅ Final Answers:
- Amount of solvent needed: 200 mL of toluene
- Amount of A recovered: 16 g
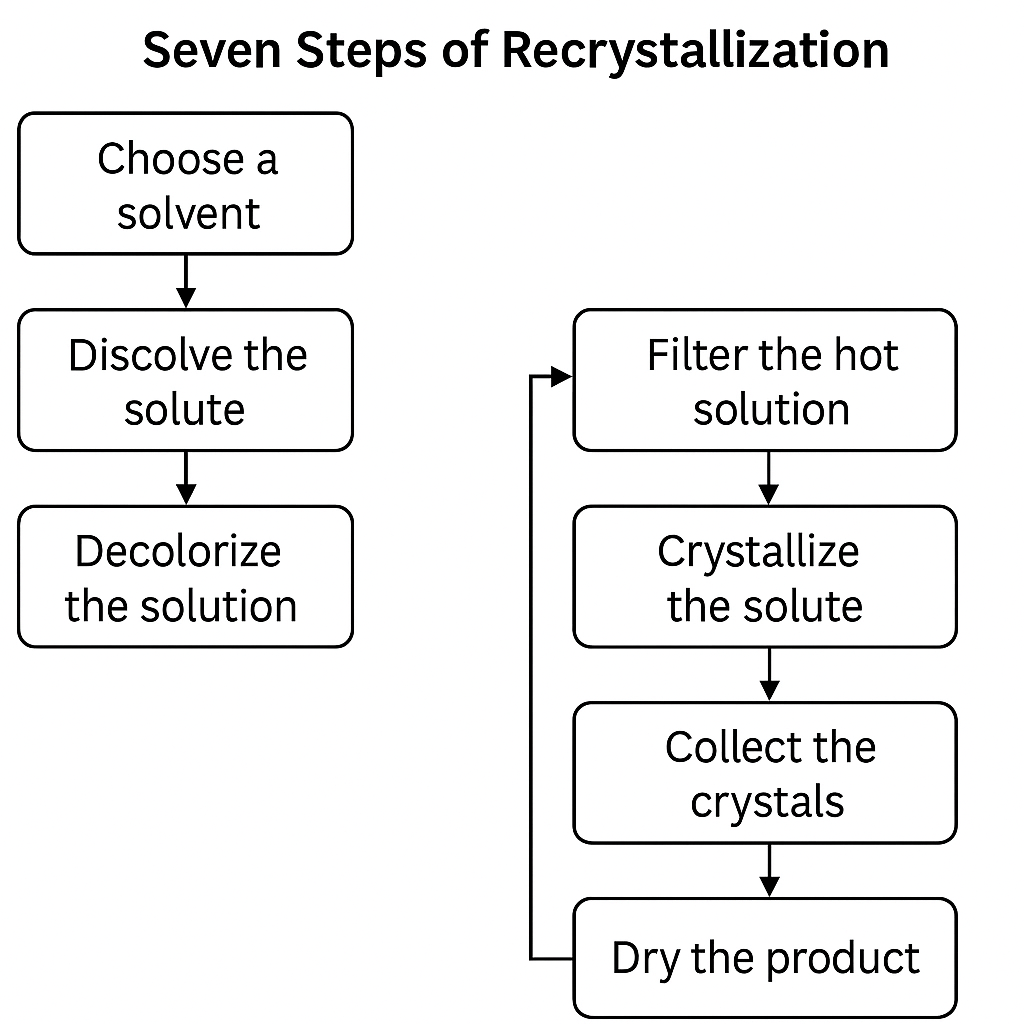
Question 4: Seven Steps of Recrystallization
- Choose a solvent – should dissolve compound when hot, not when cold.
- Dissolve the solute – heat mixture in minimal hot solvent.
- Decolorize the solution – (optional) add charcoal if colored impurities exist.
- Filter the hot solution – remove insoluble impurities (hot gravity filtration).
- Crystallize the solute – cool the filtrate slowly to form crystals.
- Collect the crystals – use vacuum filtration to isolate solid.
- Dry the product – remove any residual solvent to obtain pure compound.