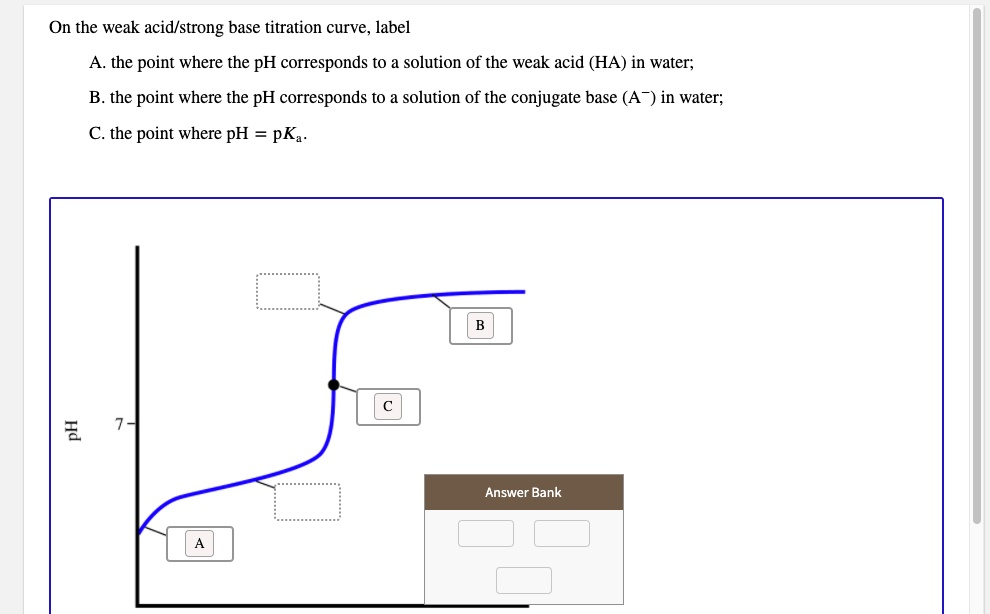
On the weak acid/strong base titration curve, label A. the point where the pH corresponds to solution of the weak acid (HA) in water; B. the point where the pH corresponds to a solution of the conjugate base (A-) in water; the point where pH = pK . 3 Answer Bank
The Correct Answer and Explanation is:
Based on the principles of acid-base titration, here are the correct labels for the specified points on the weak acid/strong base titration curve.
Correct Answer:
- A. The point where the pH corresponds to a solution of the weak acid (HA) in water: This is the initial point of the titration curve, located on the y-axis where the volume of added base is zero.
- B. The point where the pH corresponds to a solution of the conjugate base (A⁻) in water: This is the equivalence point, which is the point of inflection located at the center of the steepest section of the curve.
- C. The point where pH = pKₐ: This is the half-equivalence point, located in the middle of the initial, relatively flat buffer region of the curve.
Explanation
This graph illustrates the change in pH as a strong base is gradually added to a solution of a weak acid. Each labeled point represents a distinct chemical state of the solution.
A. The solution of the weak acid (HA) in water:
This point is at the very beginning of the titration, before any strong base has been added. The x-axis represents the volume of titrant added, so this point is at x=0. The pH of the solution is determined solely by the dissociation of the weak acid (HA) in water. The starting pH is acidic (below 7) but higher than it would be for a strong acid of the same concentration because the weak acid only partially ionizes.
B. The solution of the conjugate base (A⁻) in water:
This occurs at the equivalence point of the titration. At this point, the moles of strong base added are stoichiometrically equal to the initial moles of the weak acid. All of the weak acid (HA) has been neutralized and converted into its conjugate base (A⁻). The solution now contains primarily the salt of the conjugate base. This conjugate base is weak and will react with water in a process called hydrolysis (A⁻ + H₂O ⇌ HA + OH⁻), producing hydroxide ions. This makes the solution basic, so the pH at the equivalence point for a weak acid-strong base titration is always greater than 7. This point is identified graphically as the midpoint of the steepest vertical region of the curve.
C. The point where pH = pKₐ:
This special point is the half-equivalence point. It is reached when exactly half of the initial weak acid has been neutralized by the strong base. At this moment, the concentration of the remaining weak acid [HA] is equal to the concentration of the conjugate base [A⁻] that has been formed. The relationship between pH, pKₐ, and these concentrations is given by the Henderson-Hasselbalch equation: pH = pKₐ + log([A⁻]/[HA]). When [HA] equals [A⁻], the ratio is 1, and the logarithm of 1 is 0. The equation simplifies to pH = pKₐ. This point lies in the center of the flattest portion of the curve, known as the buffer region, where the pH resists large changes
