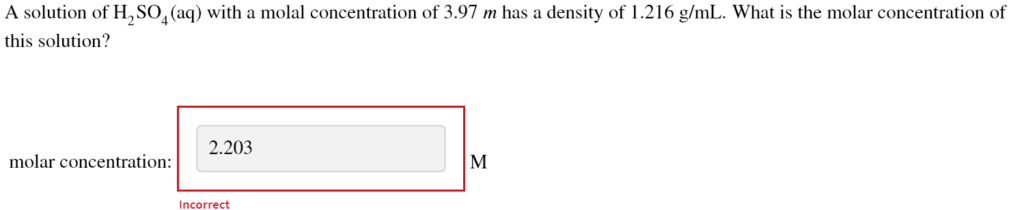
A solution of H2SO4(aq) with a molal concentration of 3.97 m has a density of 1.216 g/mL.
The Correct Answer and Explanation is:
Correct Answer: 3.47
To convert a solution’s molal concentration (molality) to its molar concentration (molarity), a systematic approach is required that utilizes the solution’s density. Molality is defined as moles of solute per kilogram of solvent, while molarity is moles of solute per liter of solution. The key is to find the total volume of the solution that contains a known amount of solute.
Let’s begin by assuming a convenient basis. Since molality is 3.97 m, it is practical to assume we have exactly 1 kilogram (1000 g) of the solvent, which is water in this aqueous solution. This means that in our 1 kg of water, there are 3.97 moles of sulfuric acid (H₂SO₄).
Next, we need the total mass of the solution, which is the mass of the solute plus the mass of the solvent. We already have the mass of the solvent (1000 g). We can find the mass of the solute by multiplying its moles by its molar mass. The molar mass of H₂SO₄ is calculated as (2 × 1.008) + 32.06 + (4 × 16.00) = 98.08 g/mol .
Mass of H₂SO₄ = 3.97 mol × 98.08 g/mol = 389.4 g.
Total mass of the solution = 1000 g (solvent) + 389.4 g (solute) = 1389.4 g.
Now, we can use the given density (1.216 g/mL) to find the total volume of the solution.
Volume = Mass / Density
Volume = 1389.4 g / 1.216 g/mL = 1142.6 mL.
Molarity is expressed in moles per liter, so we must convert the volume from milliliters to liters:
1142.6 mL ÷ 1000 mL/L = 1.1426 L.
Finally, we can calculate the molarity by dividing the moles of solute by the volume of the solution in liters:
Molarity = 3.97 mol / 1.1426 L = 3.474 M.
Rounding to three significant figures, consistent with the given molality, the molar concentration is 3.47 M.
