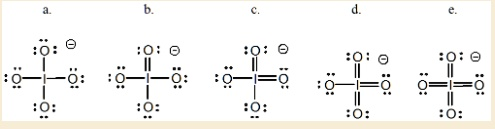
The Correct Answer and Explanation is:
The correct answer is d.
This choice represents the most stable and accurate Lewis structure for the periodate ion (IO₄⁻). The process of determining the best structure involves calculating valence electrons, arranging the atoms, and then minimizing formal charges.
First, we calculate the total number of valence electrons for the ion. Iodine (I), in Group 17, contributes 7 valence electrons. Each of the four oxygen (O) atoms, in Group 16, contributes 6 valence electrons. The ion has a negative one charge, which adds one more electron. The total is 7 + (4 × 6) + 1 = 32 valence electrons.
If we draw a structure with only single bonds connecting the central iodine to the four oxygen atoms and give every atom an octet (structure a), we fulfill the octet rule for all atoms. However, calculating the formal charges reveals a highly unstable arrangement. Each singly bonded oxygen has a formal charge of -1, while the central iodine has a formal charge of +3. A good Lewis structure should have formal charges as close to zero as possible.
To minimize these formal charges, we can create double bonds between the central iodine and the surrounding oxygens. Iodine is in the fifth period of the periodic table, which means it can have an expanded octet, accommodating more than eight electrons in its valence shell.
By forming three double bonds and leaving one single bond, as shown in structure d, we achieve the most stable distribution of formal charges. In this structure, the three double bonded oxygen atoms have a formal charge of 0. The one singly bonded oxygen atom has a formal charge of -1. The central iodine atom, now with seven bonds (three double, one single), has a formal charge of 0. This arrangement successfully minimizes formal charges and places the required overall -1 charge on one of the highly electronegative oxygen atoms, which is the most favorable location. Structures like ‘c’ and ‘e’ are less stable because they either have higher formal charges or place a negative charge on the less electronegative iodine atom.
