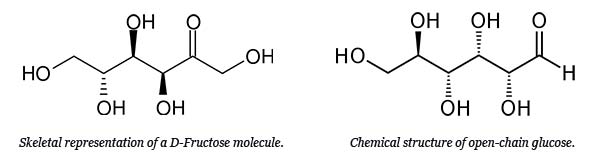
Chemical formula and image of glucose and fructose molecule that forms the high fructose corn syrup
The Correct Answer and Explanation is:
Glucose and fructose are simple sugars that are both monosaccharides and can combine to form high-fructose corn syrup (HFCS). Here’s a detailed explanation:
Chemical Formulas:
- Glucose (C₆H₁₂O₆) – A six-carbon sugar, commonly referred to as blood sugar, which is an essential energy source in living organisms.
- Fructose (C₆H₁₂O₆) – Another six-carbon sugar, often referred to as fruit sugar, naturally found in fruits, honey, and root vegetables.
How They Form High Fructose Corn Syrup:
High fructose corn syrup is made by processing corn starch to break it down into glucose. Some of this glucose is then converted into fructose by enzymes. The typical types of HFCS used in food products are HFCS-42 (42% fructose) and HFCS-55 (55% fructose). The higher concentration of fructose makes it sweeter than regular glucose.
Chemical Reaction:
When glucose and fructose combine, they form sucrose (table sugar), which is disaccharide composed of one glucose molecule and one fructose molecule. However, in HFCS, the combination of glucose and fructose doesn’t directly result in sucrose. Instead, they remain separate molecules in a syrup form, with the ratio of glucose to fructose varying.
Why HFCS is Used:
HFCS is often used in processed foods and beverages because it is cheaper than sucrose (table sugar) and has similar sweetness properties. It also has better solubility in liquids, which is why it’s commonly used in sodas, sauces, and other sweetened products.