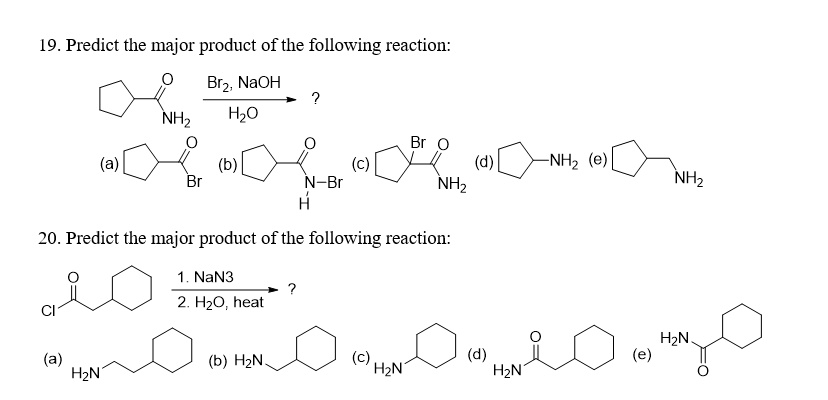
Predict the major product of the following reaction: Br
, NaOH H
O ? NH
(a) Br (b) Br O (c) (d) -NH
(e) N-Br H NH
NH
- Predict the major product of the following reaction: CI 1. NaN
- H
O, heat (a) (b) H
N H
N (c) H
N (d) H
N (e)
The Correct Answer and Explanation is:
19. Correct Answer: (d)
20. Correct Answer: (b)
Explanation:
Question 19: The reaction shown is the Hofmann rearrangement. This reaction specifically converts a primary amide (a compound with a -CONH2 group) into a primary amine (-NH2) containing one less carbon atom. The reagents, bromine (Br2) and a strong base like sodium hydroxide (NaOH), are characteristic of this transformation.
The mechanism involves several steps. First, the base deprotonates the amide. Then, the amide anion reacts with bromine to form an N-bromoamide. A second deprotonation by the base creates an anion that undergoes the key rearrangement step: the alkyl group (the cyclopentyl ring) migrates from the carbonyl carbon to the nitrogen atom, kicking out a bromide ion. This forms an isocyanate intermediate (cyclopentyl-N=C=O). In the presence of water, this isocyanate is hydrolyzed to an unstable carbamic acid, which quickly loses carbon dioxide (decarboxylates) to form the final product. The net result is the conversion of cyclopentanecarboxamide to cyclopentylamine, with the loss of the carbonyl group. Thus, the correct answer is (d).
Question 20: This reaction is a Curtius rearrangement. Similar to the Hofmann rearrangement, it is a method for producing a primary amine from a carboxylic acid derivative, also with the loss of one carbon atom. The starting material is an acyl chloride.
In the first step, the acyl chloride (2-cyclohexylacetyl chloride) reacts with sodium azide (NaN3) to form an acyl azide. Upon heating, this acyl azide is thermally unstable and rearranges. It loses a molecule of nitrogen gas (N2), a very stable leaving group, while the alkyl group (the cyclohexylmethyl group) migrates from the carbonyl carbon to the nitrogen. This creates an isocyanate intermediate (cyclohexylmethyl-N=C=O). In the second step, heating with water hydrolyzes the isocyanate, causing decarboxylation and forming the primary amine. Since the migrating group is the cyclohexylmethyl group (-CH2-cyclohexyl), the final product is cyclohexylmethanamine. Therefore, the correct answer is (b).
