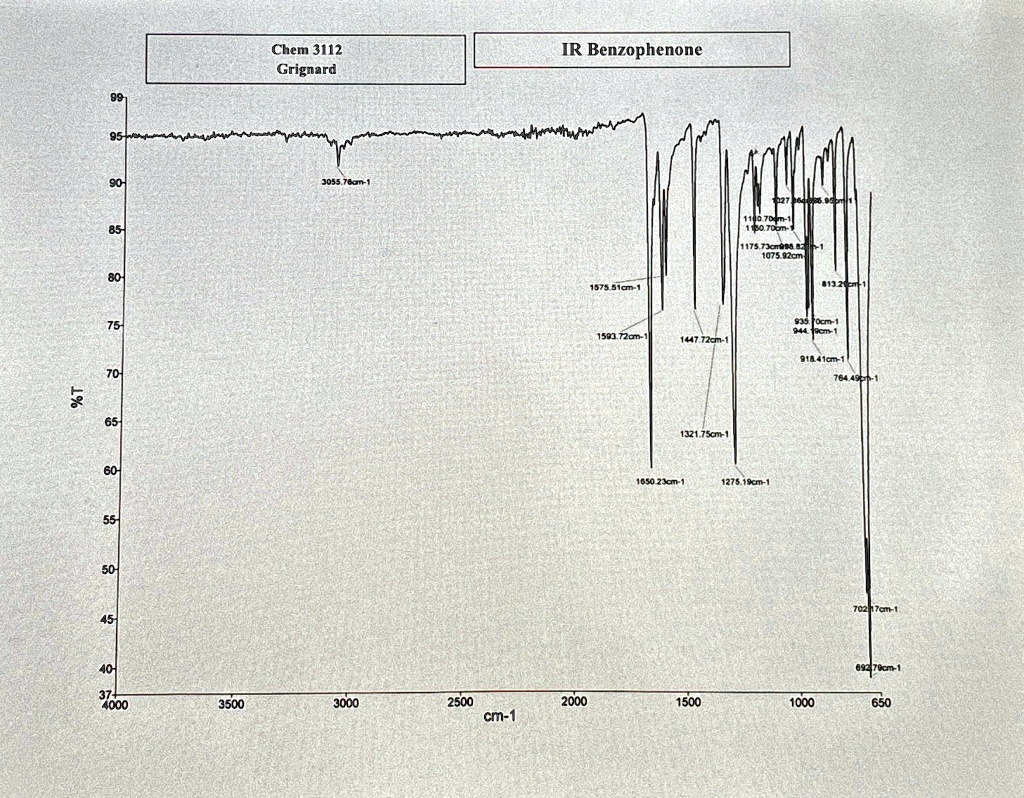
Fully annotate the IR spectra for benzophenone. Chem 3112 Grignard IR Benzophenone $6 90 85 175.7 1075.92CT 80 1575.51cm- 75 93 1593.72cm 1447.72cm 70 764 0% 65 1321.75cm 60 1650.23cm-1 1275.19cm-1 55 50 45 7027cm 40 3500 3000 2500 cm-1 2000 1500 1000 650
The Correct Answer and Explanation is:
Here is the annotation of the major peaks in the IR spectrum of benzophenone:
- 3055.76 cm⁻¹: Aromatic C-H stretch (sp² C-H)
- 1650.23 cm⁻¹: C=O stretch (conjugated ketone)
- 1593.72 cm⁻¹, 1575.51 cm⁻¹, 1447.72 cm⁻¹: Aromatic C=C ring stretching
- 1321.75 cm⁻¹, 1275.19 cm⁻¹: Ph-C(O)-Ph stretch and C-H in-plane bending
- 764.49 cm⁻¹, 692.79 cm⁻¹: Aromatic C-H out-of-plane (OOP) bending for monosubstituted ring
Explanation of the Spectrum
The provided infrared spectrum is fully consistent with the structure of benzophenone, (C₆H₅)₂CO. A detailed analysis of the absorption peaks confirms the presence of its key functional groups.
The region above 3000 cm⁻¹ is crucial for identifying C-H bonds. The peak observed at 3055.76 cm⁻¹ is characteristic of C-H stretching vibrations where the carbon atom is sp² hybridized. This confirms the presence of the aromatic rings in benzophenone. The absence of any significant peaks in the 2850-3000 cm⁻¹ range correctly indicates that there are no aliphatic (sp³ C-H) groups in the molecule.
The most intense and diagnostically significant peak in the spectrum is the sharp absorption at 1650.23 cm⁻¹. This signal is assigned to the C=O (carbonyl) stretching vibration of the ketone functional group. For a simple, non-conjugated ketone, this peak would typically appear around 1715 cm⁻¹. The lower frequency observed here is due to resonance; the carbonyl group is conjugated with both phenyl rings. This electron delocalization weakens the C=O double bond, requiring less energy to stretch, and thus lowers its absorption frequency.
The series of sharp absorptions in the 1600-1450 cm⁻¹ region, specifically at 1593.72 cm⁻¹, 1575.51 cm⁻¹, and 1447.72 cm⁻¹, are characteristic of C=C stretching vibrations within the aromatic rings, often referred to as ring breathing modes.
Finally, the fingerprint region below 1000 cm⁻¹ provides evidence for the substitution pattern of the aromatic rings. Benzophenone contains two monosubstituted phenyl rings. The two strong peaks at 764.49 cm⁻¹ and 692.79 cm⁻¹ correspond to the C-H out-of-plane (OOP) bending vibrations that are highly characteristic of monosubstitution on a benzene ring. The absence of a broad O-H stretch (3200-3600 cm⁻¹) further confirms the identity and purity of the benzophenone product, especially in the context of a Grignard reaction where an alcohol could be a byproduct.
