Testbank, Question 181: Based upon the following energy diagram, is this reaction an E1 or an E2 elimination?
The Correct Answer and Explanation is:
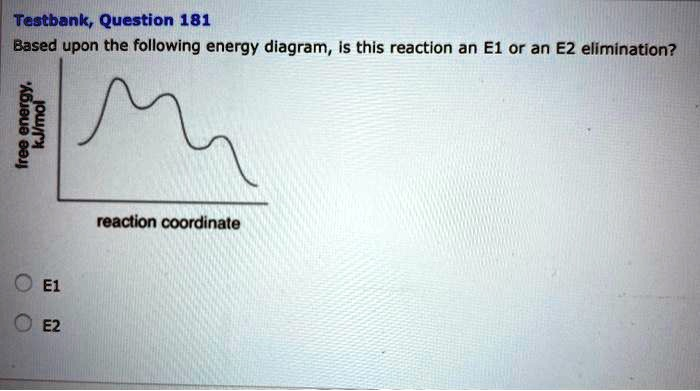
The correct answer is E1.
An energy diagram, also known as a reaction coordinate diagram, visually represents the change in free energy of a system as it transforms from reactants to products. The peaks on the diagram represent high-energy transition states, while the valleys represent more stable species like reactants, intermediates, or products. The number of peaks and valleys reveals the number of steps in a reaction mechanism.
The provided diagram shows a reaction pathway with two distinct energy peaks and one valley situated between them. This profile is characteristic of a two-step reaction mechanism. The valley represents a reaction intermediate, which is a species that is formed in one elementary step and consumed in a subsequent step.
Let’s compare this to the mechanisms of E1 and E2 elimination reactions.
The E1 (Elimination, Unimolecular) reaction is a two-step process. In the first step, the leaving group departs from the substrate to form a carbocation intermediate. This is typically the slow, rate-determining step, corresponding to the first, and usually higher, energy barrier on the diagram. The valley represents this carbocation intermediate. In the second step, a weak base removes a proton from a carbon adjacent to the carbocation, leading to the formation of an alkene. This step has its own, separate transition state, represented by the second peak on the diagram.
The E2 (Elimination, Bimolecular) reaction, on the other hand, is a concerted, one-step process. In an E2 reaction, the base removes a proton, the carbon-hydrogen bond breaks, the new pi bond forms, and the leaving group departs all at the same time through a single transition state. Consequently, the energy diagram for an E2 reaction would display only one energy peak and would not feature any intermediates.
Since the energy diagram in the question clearly shows two transition states and one intermediate, it corresponds to a two-step mechanism. Therefore, the reaction must be an E1 elimination.
