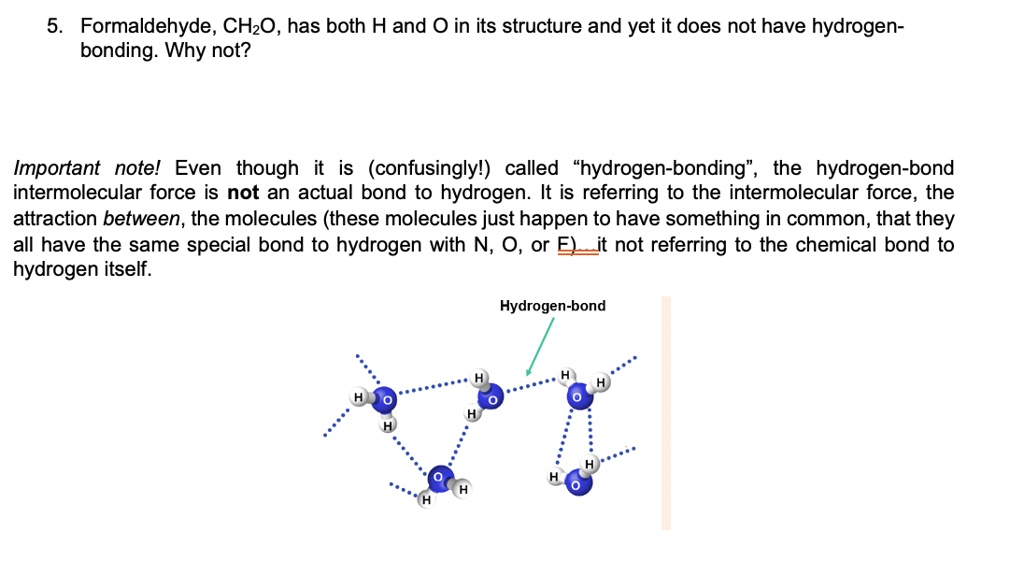
Formaldehyde, CH2O, has both H and O in its structure and yet it does not have hydrogen bonding: Why not? Important note: Even though it is (confusingly) called “hydrogen bonding”, the hydrogen bond intermolecular force is not an actual bond to hydrogen. It is referring to the intermolecular force, the attraction between the molecules (these molecules just happen to have something in common, that they all have the same special bond to hydrogen with N, O, or F). It is not referring to the chemical bond to hydrogen itself.
The Correct Answer and Explanation is:
Correct Answer:
Formaldehyde does not exhibit hydrogen bonding because for this type of intermolecular force to occur, a hydrogen atom must be directly bonded to a highly electronegative atom like nitrogen, oxygen, or fluorine within the molecule. In the structure of formaldehyde (CH₂O), the hydrogen atoms are bonded to the carbon atom, not to the oxygen atom.
Explanation
Hydrogen bonding is a particularly strong type of intermolecular force, an attraction that occurs between separate molecules. The criteria for a substance to exhibit hydrogen bonding are very specific. A molecule must possess a hydrogen atom that is covalently bonded to one of three highly electronegative elements: nitrogen (N), oxygen (O), or fluorine (F).
This H-N, H-O, or H-F bond is extremely polar. The high electronegativity of N, O, or F causes the shared electrons in the bond to be pulled strongly away from the hydrogen atom. This leaves the hydrogen with a significant partial positive charge (δ+) and the N, O, or F atom with a significant partial negative charge (δ⁻). The hydrogen bond is the electrostatic attraction between this highly positive hydrogen atom on one molecule and a lone pair of electrons on the negative N, O, or F atom of a neighboring molecule.
If we examine the molecular structure of formaldehyde, CH₂O, we find that the central carbon atom forms a double bond with the oxygen atom and single bonds with each of the two hydrogen atoms. Crucially, there are no hydrogen atoms directly attached to the oxygen atom. All hydrogens are bonded to carbon.
The carbon-hydrogen (C-H) bond is not sufficiently polar to create the conditions for hydrogen bonding. Carbon and hydrogen have relatively similar electronegativity values, so the hydrogen atom in a C-H bond does not develop the large partial positive charge required to be strongly attracted to an oxygen atom on another molecule. While the C=O bond in formaldehyde is polar, making the molecule polar overall and allowing for dipole-dipole interactions, it lacks the essential H-O covalent bond. Therefore, despite containing both hydrogen and oxygen, formaldehyde cannot form hydrogen bonds.
