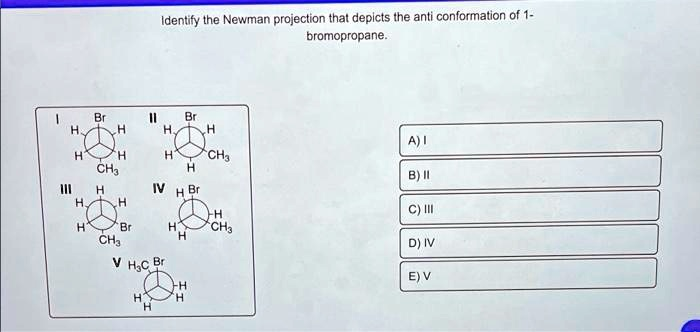
Identify the Newman projection that depicts the anti conformation of 1- bromopropane. 1 Br 11 Br H H H H A) I H H H CH
CH
H B) II III H IV H Br H H C) III H Br H CH
CH
H D) IV V H
C Br H ?? E) V H
The Correct Answer and Explanation is:
front carbon has one Br and two H atoms. The back carbon has one CH₃ and two H atoms. The Br atom is positioned directly opposite the CH₃ group (a 180° dihedral angle). This perfectly matches the definition of the anti conformation of 1-bromopropane.
* Structure II: This is also a staggered conformation of 1-bromopropane. However, the Br group and the CH₃ group are adjacent, with a dihedral angle of 60°. This is known as the “gauche” conformation, which is less stable than the anti conformation.
* Structure III: The front carbon has three H atoms, and the back carbon has a Br, a CH₃, and an H. This represents 2-bromopropane (CH₃CH(Br)CH₃), not 1-bromopropane.
* Structure IV: This is an “eclipsed” conformation of 1-bromopropane. The substituents on the front and back carbons are aligned, causing maximum steric strain. This is a high-energy, unstable conformation.
* Structure V: The front carbon has a Br, a CH₃, and an H. This also represents 2-bromopropane.
Therefore, structure I is the only Newman projection that correctly depicts the stable anti conformation of
1-bromopropane.
