Rank the following compounds in order of decreasing acid strength.
Rank the acids from the strongest to weakest. To rank items as equivalent, overlap them.
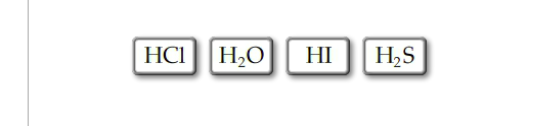
The Correct Answer and Explanation is :
Correct Ranking:
HClO₄ > HClO₃ > HClO₂ > HClO
Explanation:
The ranking of acids is based on their acid strength, which is determined by the ability of the molecule to donate a proton (H⁺) in water. This is largely influenced by the stability of the conjugate base that is formed after proton donation. For oxoacids of chlorine (HClOₓ), the acid strength increases as the number of oxygen atoms bonded to the central chlorine atom increases. Here’s why:
- Electronegativity of Oxygen: Oxygen is highly electronegative and draws electron density away from the chlorine atom. This delocalizes the negative charge on the conjugate base (ClOₓ⁻), stabilizing it. More oxygen atoms mean greater charge delocalization and stabilization of the conjugate base.
- Inductive Effect: Each additional oxygen atom enhances the inductive effect, pulling electron density away from the O-H bond, making the bond weaker and more prone to proton donation.
- Relative Acidity of HClO₄ vs. HClO₃ vs. HClO₂ vs. HClO:
- HClO₄ (Perchloric Acid): Contains 4 oxygen atoms. The negative charge on the conjugate base (ClO₄⁻) is highly delocalized over all the oxygen atoms, making it the strongest acid.
- HClO₃ (Chloric Acid): Contains 3 oxygen atoms. The conjugate base (ClO₃⁻) is less stabilized than ClO₄⁻, making HClO₃ weaker than HClO₄.
- HClO₂ (Chlorous Acid): Contains 2 oxygen atoms. Its conjugate base (ClO₂⁻) is less stabilized than ClO₃⁻, making it a weaker acid than HClO₃.
- HClO (Hypochlorous Acid): Contains only 1 oxygen atom. Its conjugate base (ClO⁻) is the least stabilized, making HClO the weakest acid in this series.
In conclusion, the strength of these oxoacids of chlorine directly correlates with the number of oxygen atoms, due to the inductive and resonance effects that stabilize the conjugate base.