- Which is a more polar solvent mixture (mobile phase) for TLC, 1:1 ethyl acetate/hexane or 4:1 ethyl acetate/hexane?
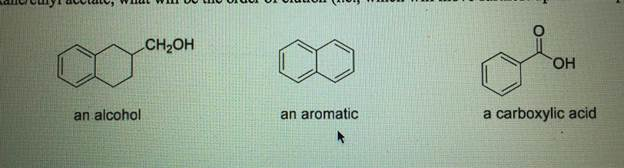
- The following three compounds are spotted on a TLC plate. The TLC plate is developed using 7:3 hexane/ethyl acetate, what will be the order of elution (i.e., which will move furthest up the TLC plate)?
3. Why must the spots applied to a TLC plate be above the level of the developing solvent?
The Correct Answer and Explanation is :
1. Which is a more polar solvent mixture (mobile phase) for TLC, 1:1 ethyl acetate/hexane or 4:1 ethyl acetate/hexane?
The 4:1 ethyl acetate/hexane mixture is more polar than the 1:1 mixture. This is because ethyl acetate is more polar than hexane. In a 4:1 mixture, the proportion of ethyl acetate is higher, which increases the polarity of the mobile phase compared to the 1:1 mixture, where both solvents are in equal amounts.
In TLC, the polarity of the mobile phase is crucial because it influences how compounds move up the plate. More polar solvents will tend to move more polar compounds higher up the plate, while less polar solvents will cause nonpolar compounds to travel further.
2. The following three compounds are spotted on a TLC plate. The TLC plate is developed using 7:3 hexane/ethyl acetate. What will be the order of elution?
For this, the order of elution depends on the polarity of the compounds. The most polar compound will interact more strongly with the stationary phase (usually silica gel) and thus move more slowly up the TLC plate, while the least polar compound will move faster as it interacts less with the stationary phase and more with the mobile phase.
Since the solvent mixture is 7:3 hexane/ethyl acetate, the mobile phase is relatively non-polar (hexane) with a small amount of polar solvent (ethyl acetate). This means that compounds with lower polarity will move faster up the plate.
Based on the relative polarities of the compounds, the least polar compound will move furthest up the TLC plate, followed by the moderately polar compound, and finally, the most polar compound will move the least.
Without the image, I can hypothesize that the order of elution would follow this general pattern. If you provide more details on the specific structures of the compounds, I can give a more accurate answer.
3. Why must the spots applied to a TLC plate be above the level of the developing solvent?
The spots applied to a TLC plate must be above the level of the developing solvent to prevent the compounds from dissolving directly into the solvent during the initial stage of development. If the spots were submerged in the solvent, the compounds would simply dissolve into the solvent instead of migrating up the TLC plate. This would render the experiment useless because no separation would occur.
The stationary phase (usually silica gel) on the TLC plate interacts with the compounds, causing them to move upward as the solvent travels up the plate by capillary action. When the solvent reaches the spots, it begins to carry the compounds upward according to their affinity for the mobile phase and the stationary phase. If the spots were submerged, they would just be swept into the mobile phase, making it impossible to observe the separation based on polarity.
Additionally, if the spots are too close to the solvent level, the compounds could be prematurely dissolved and run together, leading to poor resolution or inaccurate results. The optimal way to spot compounds is to apply them above the solvent level so that, during development, they gradually move up the plate, separating based on their interaction with both the solvent and the stationary phase.