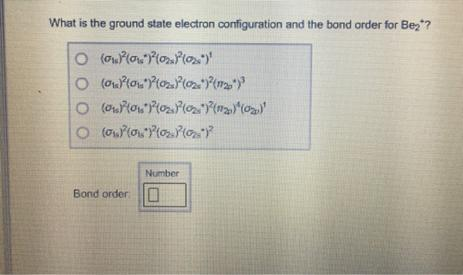
What is the ground state electron configuration and the bond order for Be2? Number Bond order:
The Correct Answer and Explanation is :
The ground state electron configuration of the beryllium dimer (Be₂) and its bond order can be determined using molecular orbital (MO) theory.
Electron Configuration of Be₂:
Each beryllium (Be) atom has an atomic number of 4, with the electron configuration 1s² 2s². In the Be₂ molecule, the 2s atomic orbitals from each atom combine to form bonding (σ₂s) and antibonding (σ₂s) molecular orbitals. The 1s orbitals also combine to form bonding (σ₁s) and antibonding (σ₁s) molecular orbitals. Given that Be₂ has a total of 8 electrons (4 from each Be atom), the electron configuration in molecular orbitals is:
- σ₁s² σ₁s² σ₂s² σ₂s²
This configuration indicates that all electrons occupy bonding and antibonding molecular orbitals formed from the 1s and 2s atomic orbitals.
Bond Order of Be₂:
Bond order is calculated using the formula:
[ \text{Bond Order} = \frac{(\text{Number of electrons in bonding MOs}) – (\text{Number of electrons in antibonding MOs})}{2} ]
In Be₂, the bonding electrons are 4 (2 from σ₁s and 2 from σ₂s), and the antibonding electrons are also 4 (2 from σ₁s and 2 from σ₂s). Therefore, the bond order is:
[ \text{Bond Order} = \frac{4 – 4}{2} = 0 ]
A bond order of zero suggests that Be₂ does not have a stable bond under normal conditions.
Explanation:
According to molecular orbital theory, the stability of a molecule depends on the difference between the number of electrons in bonding and antibonding molecular orbitals. In the case of Be₂, the equal number of electrons in bonding and antibonding orbitals results in a bond order of zero, indicating no net bonding interaction between the two beryllium atoms. This theoretical prediction aligns with the fact that Be₂ is not a stable molecule under standard conditions.
However, experimental data has shown that Be₂ can exist transiently under specific conditions, such as in a gas phase at very low temperatures. This suggests that while the bond order calculation predicts instability, Be₂ can form under certain circumstances, albeit with a very weak bond.
In summary, the ground state electron configuration of Be₂ is σ₁s² σ₁s² σ₂s² σ₂s², and its bond order is zero, indicating no stable bonding under normal conditions.