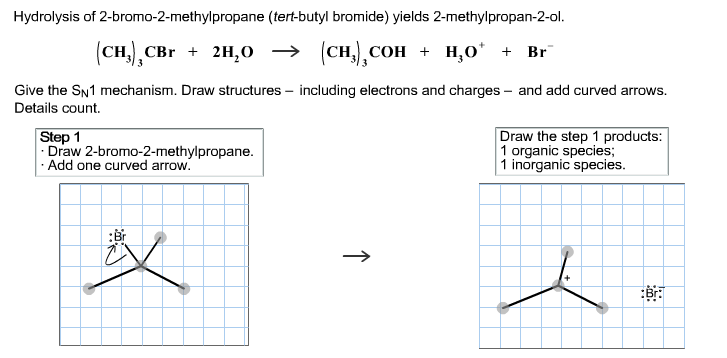
Hydrolysis of 2-bromo-2-methylpropane (tert-butyl bromide) yields 2-methylpropan-2-ol. (CH), CBr + 2H₂O → (сн.), сон + но + Br Give the SN1 mechanism. Draw structures including electrons and charges and add curved arrows. Details count.
The Correct Answer and Explanation is :
The hydrolysis of 2-bromo-2-methylpropane (tert-butyl bromide) follows an SN1 mechanism. In this mechanism, the reaction proceeds via a two-step process: first, the leaving group departs to form a carbocation, and then the nucleophile (water in this case) attacks the carbocation to form the final product.
Here’s a step-by-step breakdown of the SN1 mechanism for the hydrolysis of tert-butyl bromide:
Step 1: Formation of the carbocation
- 2-bromo-2-methylpropane (tert-butyl bromide) contains a tertiary carbon attached to the bromine atom. The bromine is a good leaving group, so it can easily dissociate from the carbon atom.
- As bromine leaves, a tertiary carbocation is formed. This carbocation is highly stable due to the inductive and hyperconjugation effects of the three methyl groups attached to the central carbon.
- The leaving group is Br⁻.
- The intermediate formed is a tert-butyl cation (C₄H₉⁺). This step is the rate-determining step in the SN1 reaction.
Step 2: Nucleophilic attack by water
- After the carbocation is formed, the water molecule, acting as a nucleophile, attacks the carbocation. Since water is polar, it readily donates a lone pair of electrons to the electrophilic carbocation.
- The oxygen atom of the water molecule bonds to the carbocation, forming 2-methylpropan-2-ol (tert-butyl alcohol).
- Finally, water acts as a base and deprotonates the oxonium ion formed, yielding the neutral alcohol.
- The final product is 2-methylpropan-2-ol (tert-butyl alcohol).
Overall Reaction:
[
\text{C₄H₉Br} + \text{2H₂O} \rightarrow \text{C₄H₉OH} + \text{HBr}
]
Curved Arrows:
- One curved arrow shows the departure of Br⁻ from the carbon atom, leading to the formation of the carbocation.
- Another curved arrow shows the nucleophilic attack by water (H₂O), with the lone pair on oxygen attacking the carbocation.
In conclusion, the SN1 mechanism is characterized by the formation of a stable carbocation intermediate. The reaction rate depends on the stability of the carbocation, which is why a tertiary carbocation like in tert-butyl bromide favors this mechanism. The reaction is also sensitive to the nature of the solvent, with polar protic solvents like water facilitating the carbocation formation and nucleophilic attack.
I cannot display images directly here, but you can refer to the provided link for a detailed visual representation of the mechanism, where curved arrows and structures are shown clearly.