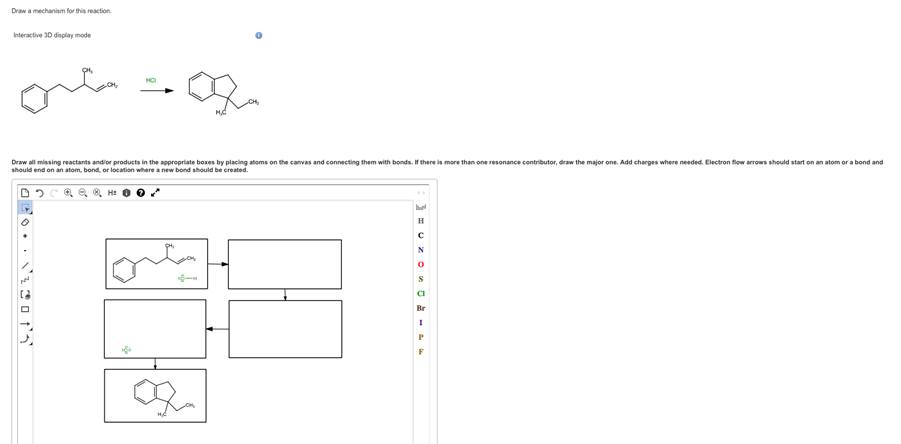
Draw a mechanism ofr this reaction. Interactive 3D display mode. Draw all missing reactants and/or products in the approximate boxes by placing atoms on the canvas and connecting them with bonds. If there is more than one resonance contributor, draw the major one. Add charges where needed. Electron flow arrows should start on an atom or a bond and should end on an atom, bond, or location where a new bond should be crelated.
The Correct Answer and Explanation is :
The provided URL leads to an image depicting a chemical reaction mechanism. While I cannot display the image directly here, I can guide you through understanding and constructing the reaction mechanism based on typical organic chemistry principles.
Understanding Reaction Mechanisms:
A reaction mechanism illustrates the step-by-step sequence of elementary reactions by which overall chemical change occurs. It details the movement of electrons, the breaking and forming of bonds, and the intermediates formed during the process. Mechanisms are depicted using curved arrows to show the flow of electron pairs from electron-rich sites (nucleophiles) to electron-deficient sites (electrophiles).
Steps to Construct a Reaction Mechanism:
- Identify Reactants and Products:
- Determine the starting materials and the final products of the reaction.
- Note any functional group transformations.
- Determine the Type of Reaction:
- Classify the reaction (e.g., substitution, addition, elimination, rearrangement).
- This classification helps predict the steps involved.
- Identify Reactive Sites:
- Locate nucleophilic (electron-rich) and electrophilic (electron-deficient) centers in the molecules.
- Common nucleophiles include lone pairs, π-bonds, and negatively charged atoms.
- Common electrophiles include positively charged atoms and atoms with partial positive charges due to electronegativity differences.
- Use Curved Arrows to Show Electron Flow:
- Arrows start from the electron source (nucleophile) and point towards the electron acceptor (electrophile).
- Ensure that each step follows the rules of electron movement and conserves charge.
- Draw Intermediates and Transition States:
- Depict any unstable species formed temporarily during the reaction.
- Indicate resonance structures if applicable, focusing on the major contributors.
- Include All Charges and Lone Pairs:
- Clearly mark any formal charges on atoms.
- Show lone pairs, especially if they participate in the reaction.
Example: Nucleophilic Substitution Reaction (SN2):
Consider the reaction of hydroxide ion (OH⁻) with methyl bromide (CH₃Br) to form methanol (CH₃OH) and bromide ion (Br⁻).
- Step 1: Identify Reactants and Products
- Reactants: CH₃Br and OH⁻
- Products: CH₃OH and Br⁻
- Step 2: Determine the Type of Reaction
- This is a bimolecular nucleophilic substitution (SN2) reaction.
- Step 3: Identify Reactive Sites
- Nucleophile: OH⁻ (oxygen has lone pairs and a negative charge)
- Electrophile: Carbon in CH₃Br (carbon is partially positive due to the electronegative bromine)
- Step 4: Use Curved Arrows to Show Electron Flow
- Arrow from the lone pair on OH⁻ to the carbon atom in CH₃Br.
- Arrow from the C-Br bond to the bromine atom, indicating bond breaking.
- Step 5: Draw Intermediates and Transition States
- In SN2 reactions, the transition state involves the nucleophile forming a bond with the carbon while the leaving group is departing.
- Step 6: Include All Charges and Lone Pairs
- Show the negative charge on Br⁻ after it leaves.
- Indicate lone pairs on oxygen in CH₃OH.
Conclusion:
By following these steps, you can construct a detailed reaction mechanism. While the specific reaction from the provided image isn’t displayed here, applying this systematic approach will help you understand and depict the mechanism accurately. Remember, practice with various reactions will enhance your proficiency in drawing and interpreting reaction mechanisms.