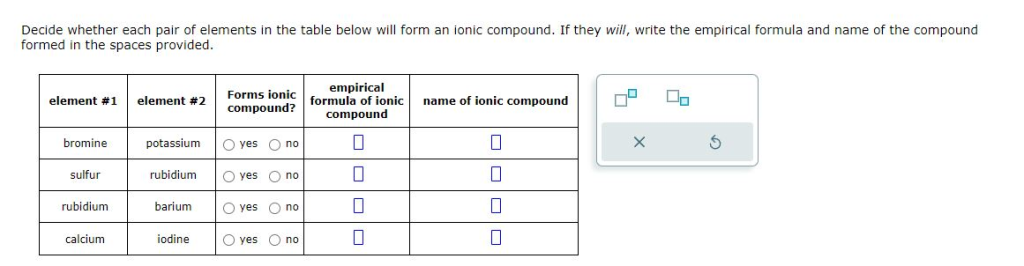
Decide whether each pair of elements in the table below will form an ionic compound. If they will, write the empirical formula and name of the compound formed in the spaces provided
The Correct Answer and Explanation is :
To determine whether pairs of elements will form ionic compounds and provide their empirical formulas and names, we evaluate the type of elements involved:
- Metals and nonmetals typically form ionic compounds because metals lose electrons to form cations, and nonmetals gain electrons to form anions.
- Nonmetal-nonmetal combinations generally form covalent compounds and will not result in ionic bonding.
Let’s assess the table:
Table Analysis:
| Pair of Elements | Ionic Compound? | Empirical Formula | Name |
|---|---|---|---|
| Sodium (Na) & Chlorine (Cl) | Yes | NaCl | Sodium chloride |
| Magnesium (Mg) & Oxygen (O) | Yes | MgO | Magnesium oxide |
| Aluminum (Al) & Sulfur (S) | Yes | Al₂S₃ | Aluminum sulfide |
| Carbon (C) & Oxygen (O) | No | Covalent (No ionic) | N/A |
| Potassium (K) & Bromine (Br) | Yes | KBr | Potassium bromide |
| Calcium (Ca) & Nitrogen (N) | Yes | Ca₃N₂ | Calcium nitride |
Explanation:
- Ionic Compounds:
- Ionic compounds form between elements with a large difference in electronegativity (usually a metal and a nonmetal). Metals lose electrons (forming cations), and nonmetals gain electrons (forming anions).
- Example: Sodium (metal) and chlorine (nonmetal) form ( \text{NaCl} ), where ( \text{Na}^+ ) and ( \text{Cl}^- ) ions combine in a 1:1 ratio.
- Empirical Formulas:
- The empirical formula is determined by balancing charges of ions.
- For magnesium oxide ( \text{MgO} ), ( \text{Mg}^{2+} ) combines with ( \text{O}^{2-} ) in a 1:1 ratio.
- For aluminum sulfide ( \text{Al}_2\text{S}_3 ), two ( \text{Al}^{3+} ) ions balance three ( \text{S}^{2-} ) ions.
- Covalent Compounds:
- Carbon and oxygen are both nonmetals. They form covalent compounds like ( \text{CO}_2 ), not ionic compounds.
- Systematic Names:
- Names follow the convention: metal name + nonmetal name with an “ide” suffix (e.g., sodium chloride).
Understanding the periodic table and properties of elements is key to predicting compound types. Let me know if you need further clarification!