Give the correct formula for aluminum sulfate.
A) Al2SO4
B) Al(SO4)3
C) Al3(SO4)2
D) Al2(SO4)3
The correct answer and explanation is :
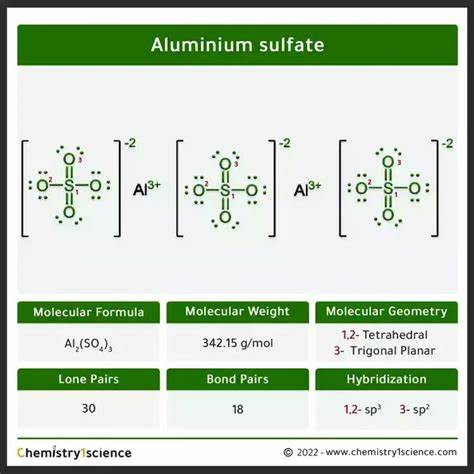
The correct formula for aluminum sulfate is D) Al₂(SO₄)₃.
Explanation:
Aluminum sulfate is an inorganic compound with the chemical formula Al₂(SO₄)₃. It is commonly used in water purification, as a mordant in dyeing, and in the production of paper. Understanding its formula involves recognizing the ionic nature of the compound and the charges of the constituent ions.
- Aluminum Ion (Al³⁺): Aluminum, as a metal in Group 13 of the periodic table, forms a cation with a 3+ charge (Al³⁺). This means that aluminum atoms need to lose three electrons to achieve a stable electron configuration.
- Sulfate Ion (SO₄²⁻): The sulfate ion is a polyatomic ion consisting of one sulfur atom covalently bonded to four oxygen atoms, with an overall charge of 2- (SO₄²⁻). This ion is negatively charged, meaning it needs to balance out the positive charges of the aluminum ions.
- Balancing the Charges: To create a neutral compound, the charges must balance. Since each aluminum ion has a +3 charge and each sulfate ion has a -2 charge, the least common multiple of 3 and 2 is 6. This means that two aluminum ions (each with a +3 charge) will balance with three sulfate ions (each with a -2 charge).
Thus, the formula for aluminum sulfate is Al₂(SO₄)₃, which ensures that the total positive charge (+6) is balanced by the total negative charge (-6), resulting in a neutral compound.
Why the other options are incorrect:
- A) Al₂SO₄: This does not balance the charges correctly.
- B) Al(SO₄)₃: This formula suggests one aluminum ion with three sulfate ions, which doesn’t balance the charges.
- C) Al₃(SO₄)₂: This also doesn’t result in a neutral charge balance.