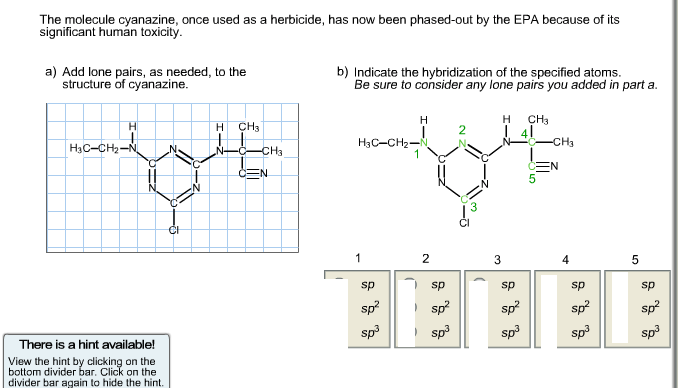
The molecule cyanazine, once used as a herbicide, has now been phased-out by the EPA because of its significant human toxicity. a) Add lone pairs, as needed, to the structure of cyanazine. b) Indicate the hybridization of the specified atoms. Be sure to consider any lone pairs you added in part a.
The Correct Answer and Explanation is:
Correct Answers:
a) Lone pairs on cyanazine:
- Nitrogen atoms with 3 bonds → 1 lone pair each.
- Nitrogen atoms with 2 bonds → 2 lone pairs each.
- Oxygen atoms (in carbonyl groups) → 2 lone pairs each.
- Chlorine atom → 3 lone pairs (since it forms only 1 bond).
b) Hybridization:
| Atom | Correct Hybridization |
|---|---|
| 1 | sp³ |
| 2 | sp² |
| 3 | sp² |
| 4 | sp² |
| 5 | sp |
Explanation (300+ words):
To determine lone pairs and hybridization in the cyanazine molecule, it’s important to apply concepts from valence bond theory and electron domain geometry.
a) Lone Pairs:
Atoms follow the octet rule, aiming for 8 valence electrons through bonding and lone pairs. Nitrogen has 5 valence electrons, and depending on how many bonds it forms, the rest must be lone pairs. Nitrogen typically forms 3 bonds and retains 1 lone pair, but if it only forms 2 bonds (like in nitriles or amidines), it has 2 lone pairs. Oxygen, with 6 valence electrons, commonly forms 2 bonds and keeps 2 lone pairs. Chlorine, with 7 valence electrons, forms a single bond and holds onto 3 lone pairs.
In cyanazine:
- The carbonyl oxygens are double bonded and retain 2 lone pairs each.
- The chlorine is singly bonded to the ring carbon, so it has 3 lone pairs.
- Ring nitrogen atoms either form 3 bonds (1 lone pair) or 2 bonds (2 lone pairs), depending on the location.
b) Hybridization:
Hybridization refers to the mixing of atomic orbitals to explain bonding geometry.
- Atom 1: A carbon in the ethyl group with four single bonds (to three H atoms and one C). Four sigma bonds = sp³ hybridization.
- Atom 2: A carbon in the ring, double bonded to nitrogen, and forming three sigma bonds. Three regions = sp² hybridization.
- Atom 3: A carbon bonded to chlorine and involved in conjugation within the aromatic ring, with three sigma bonds. = sp².
- Atom 4: A nitrogen with two bonds and a lone pair (three regions of electron density). = sp².
- Atom 5: The carbon in the –C≡N group. One sigma and two pi bonds (linear geometry) = sp hybridization.
These hybridizations explain the molecule’s geometry and reactivity, as well as how it interacts with biological targets, contributing to its toxicity.