What is the IUPAC name of the following? CH3CH2COCH3 Submit Answer Retry Entire Group 8 more group attempts remaining
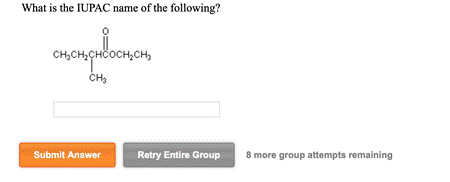
What is the IUPAC name of the following? CH3CH2CHCOCH2CH3 Submit Answer Retry Entire Group 8 more group attempts remaining
What is the IUPAC name of the following compound? H OH Submit Answer Retry Entire Group 8 more group attempts remaining
What is the IUPAC name of the following compound? CH2CH2CHC-H CH2CH3 Submit Answer Retry Entire Group 8 more group attempts remaining
The Correct Answer and Explanation is:
The structure in the image shows a molecule with the following structure:
O
||
CH3CH2–C–CH(CH3)–CH2CH3
Step-by-step IUPAC Naming:
- Identify the longest carbon chain that includes the carbonyl (C=O) group:
- The longest chain that includes the ketone is pentane (5 carbons).
- Number the chain from the end nearest to the carbonyl group:
- The carbonyl group must get the lowest possible number. So, start numbering from the left:
CH3–CH2–CO–CH(CH3)–CH3
→ Numbers: 1–2–3(C=O)–4–5
→ The carbonyl carbon is position 3.
- The carbonyl group must get the lowest possible number. So, start numbering from the left:
- Identify and name the substituents:
- There is a methyl group (CH3) attached to carbon 4.
- Combine the name:
- The parent chain is pentanone because it has 5 carbons and a ketone group.
- The methyl group is on carbon 4.
- Therefore, the IUPAC name is 4-methyl-2-hexanone.
✅ Final Answer: 4-methyl-2-hexanone
300-Word Explanation:
The IUPAC naming of organic compounds follows a systematic approach to ensure clarity and uniformity. The molecule in question contains a ketone functional group (C=O), so the compound falls under the category of ketones. Ketones are named by identifying the longest carbon chain containing the carbonyl group and replacing the “-e” ending of the alkane with “-one”.
First, identify the longest continuous chain that includes the carbonyl group. In this case, the chain has six carbon atoms, so the parent hydrocarbon is hexane. Because the carbonyl group is at position 2 (numbering from the direction that gives the ketone the lowest number), the parent name becomes hexan-2-one or more simply, 2-hexanone.
Next, we identify the substituents. There is a methyl group (–CH₃) attached to the 4th carbon of the main chain. This substituent must be included in the name and its position indicated.
Putting it all together, we place the substituent name and position as a prefix and the functional group and its position as a suffix. Therefore, the correct IUPAC name of the compound is 4-methyl-2-hexanone. The number 4 indicates the position of the methyl group, and 2 indicates the position of the ketone group.
This naming ensures that the structure of the molecule can be unambiguously determined from the name. It follows standard IUPAC conventions, including prioritizing the functional group, giving the lowest possible numbers, and listing substituents alphabetically

.