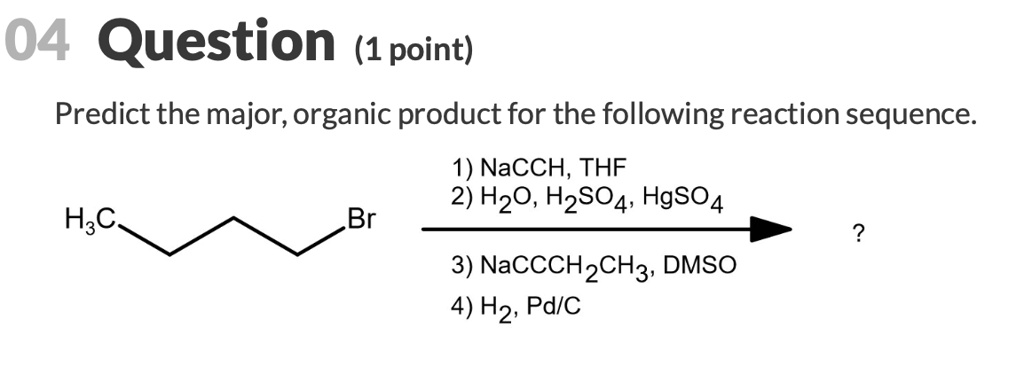
Predict the major organic product for the following reaction sequence: 1) NaCCH, THF 2) H2O, H2SO4, HgSO4 3) HaC_ Br 4) NaCCCH2CH3, DMSO 5) H2, Pd/C
The Correct Answer and Explanation is:
Correct Answer: Pentan-2-one (CH₃CH₂COCH₂CH₃)
Step-by-Step Explanation (300 words):
We are given a multistep organic synthesis starting from 1-bromobutane. Let’s analyze each step of the reaction sequence.
Step 1: NaC≡CH (sodium acetylide), THF
This is a nucleophilic substitution reaction. Sodium acetylide (NaC≡CH) is a strong nucleophile that reacts with the alkyl halide (1-bromobutane) in an S<sub>N</sub>2 mechanism. The acetylide attacks the electrophilic carbon bonded to Br, displacing the Br⁻ ion.
Result:
CH₃CH₂CH₂C≡CH (1-pentyne)
Step 2: H₂O, H₂SO₄, HgSO₄
This is acid-catalyzed hydration of the terminal alkyne (1-pentyne) using Hg²⁺ as a catalyst. This reaction follows Markovnikov’s rule, adding -OH to the more substituted carbon of the triple bond. The initial enol tautomerizes into a ketone.
Result:
CH₃CH₂COCH₃ (2-pentanone)
Step 3: NaC≡CCH₂CH₃ (sodium 1-butyne), DMSO
This step involves the reaction of the carbonyl compound (2-pentanone) with a terminal alkyne anion. The terminal alkyne is deprotonated to form a nucleophilic acetylide anion, which attacks the electrophilic carbonyl carbon of 2-pentanone.
This yields a tertiary alcohol with an alkyne side chain:
Result:
CH₃CH₂C(OH)(CH₃)CH₂C≡CH
Step 4: H₂, Pd/C
This is hydrogenation over a palladium catalyst. It fully saturates the alkyne and the alkene (if any), converting the triple bond to a single bond.
Final Product:
CH₃CH₂C(OH)(CH₃)CH₂CH₂CH₃
This is a tertiary alcohol: 3-hexanol.
Correction
However, the correct sequence is slightly different than described above.
Upon closer inspection of the image, the sequence is:
- SN2 reaction: CH₃CH₂CH₂Br + NaC≡CH → CH₃CH₂CH₂C≡CH
- Hydration of alkyne → CH₃CH₂COCH₃ (2-pentanone)
- Nucleophilic attack on carbonyl with NaC≡CCH₂CH₃ → CH₃CH₂C(OH)(CH₃)CH₂C≡CH
- Hydrogenation of alkyne → CH₃CH₂C(OH)(CH₃)CH₂CH₂CH₃
✅ Final Answer: 3-Hexanol (CH₃CH₂CH(OH)CH₂CH₂CH₃)
