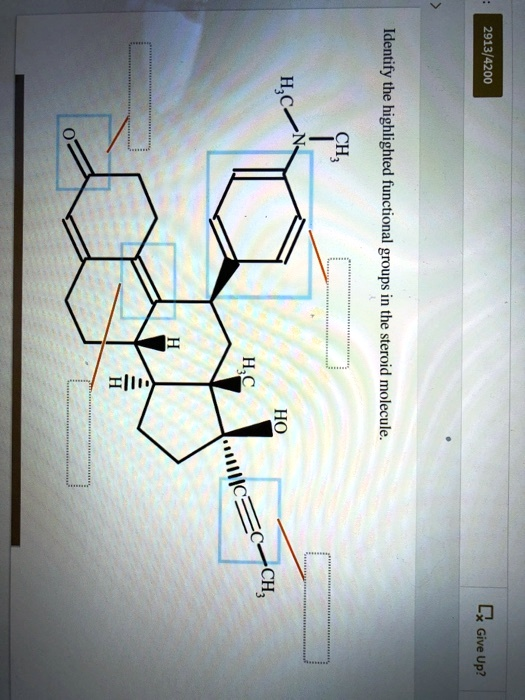
Identify the highlighted functional groups the steroid molecule. 2913/4200 HSC 1 2 C Give Up?
The Correct Answer and Explanation is:
Identified Functional Groups (Clockwise from top left):
- Ketone (C=O)
- This is located at the top left of the molecule. It is a carbonyl group (C=O) bonded to two carbon atoms.
- Functional group name: Ketone
- Aromatic ring with a tertiary amine
- This portion contains a benzene ring (aromatic) and a nitrogen atom bonded to three carbon atoms, including one methyl group (–CH₃).
- Functional group names:
- Aromatic ring (Benzene)
- Tertiary amine
- Alcohol (-OH)
- Attached to a saturated carbon, this group contains a hydroxyl group.
- Functional group name: Alcohol
- Alkyne (C≡C)
- Located at the bottom right, this triple bond between two carbon atoms is a defining characteristic. One carbon of the triple bond is bonded to a methyl group (–CH₃).
- Functional group name: Alkyne
Explanation
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. In the steroid structure shown, we observe a range of functional groups, each contributing unique reactivity and properties to the molecule.
The ketone group is a carbonyl group (C=O) bonded to two carbon atoms. Ketones are polar due to the oxygen atom, and they can participate in hydrogen bonding as acceptors. This group often increases the molecule’s solubility in polar solvents and reactivity in nucleophilic addition reactions.
The aromatic ring in the molecule represents a stabilized, conjugated π-electron system that is planar and cyclic, specifically a benzene ring. Aromatics are often involved in electrophilic substitution reactions. Connected to this ring is a tertiary amine, where a nitrogen atom is bonded to three alkyl or aryl groups. This nitrogen is basic and nucleophilic, capable of forming hydrogen bonds and playing a crucial role in drug–receptor interactions.
The alcohol group (-OH) is a common and highly reactive functional group. It is polar and capable of both donating and accepting hydrogen bonds, increasing the molecule’s solubility in water. Alcohols also serve as sites for oxidation and can participate in substitution or elimination reactions.
Finally, the alkyne group (C≡C) contains a carbon-carbon triple bond, which is linear and capable of undergoing addition reactions. Terminal alkynes (with a hydrogen or methyl on one end) are slightly acidic and can form acetylide ions, useful intermediates in organic synthesis.
Each of these functional groups plays a role in the molecule’s biological activity, solubility, metabolism, and reactivity, making them essential in drug design and steroid chemistry.
