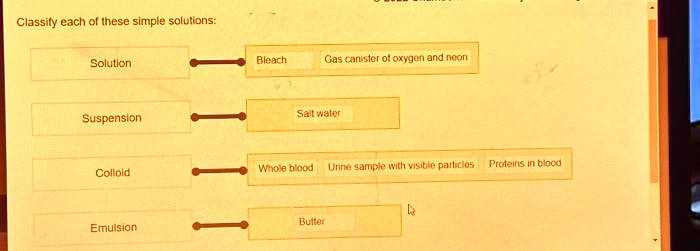
Classify each of these simple solutions: Solution Suspension Colloid Emulsion Bleach Gas canister of oxygen and neon Salt water Whole blood Urine sample with visible particles Proteins in blood Butter
The Correct Answer and Explanation is:
Here is the correct classification for each substance, followed by a detailed explanation.
Correct Answer
- Solution: Bleach, Gas canister of oxygen and neon, Salt water
- Suspension: Urine sample with visible particles
- Colloid: Whole blood, Proteins in blood
- Emulsion: Butter
Explanation
The classification of these mixtures depends on the size of the particles dispersed within them and how they behave. The four categories represent different points on a spectrum of particle size.
A solution is a homogeneous mixture where the solute particles are extremely small, typically ions or molecules. These particles dissolve completely, do not settle out over time, and cannot be separated by simple filtration. They are too small to scatter light. Bleach is a solution of sodium hypochlorite in water. Salt water is a solution of sodium chloride in water. A gas canister of oxygen and neon is also a solution because gases mix perfectly at the molecular level to form a homogeneous mixture.
A suspension is a heterogeneous mixture containing large particles that are visible to the naked eye. These particles are heavy enough to settle out over time due to gravity and can be easily separated by filtration. The “urine sample with visible particles” is a suspension because the presence of visible, undissolved solids indicates they will eventually settle.
A colloid features particles that are intermediate in size between those in solutions and suspensions. These particles are larger than single molecules but small enough to remain dispersed indefinitely without settling. They cannot be separated by simple filtration and are large enough to scatter a beam of light, a phenomenon known as the Tyndall effect. Whole blood is a classic example of a colloid because it contains red blood cells, white blood cells, and platelets dispersed in plasma. Proteins in blood also form a colloid, as these large macromolecules are suspended in the plasma.
An emulsion is a specific type of colloid formed by the mixture of two or more liquids that are normally immiscible, like oil and water. One liquid is dispersed as tiny droplets within the other. Butter is an emulsion, specifically consisting of water droplets dispersed throughout a continuous phase of milk fat
