Draw the Lewis structure for the chlorine pentafluoride (CIF
) molecule
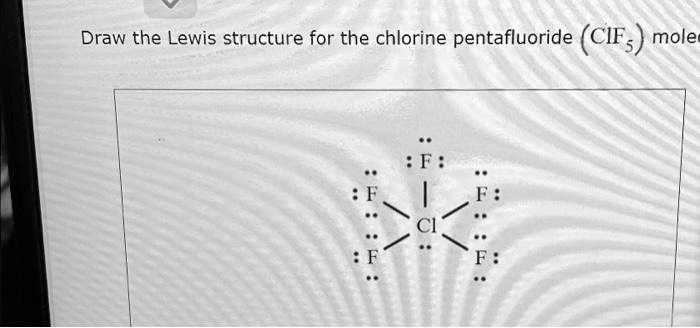
The Correct Answer and Explanation is:
Here’s the correct Lewis structure for chlorine pentafluoride (ClF₅), followed by a detailed explanation:
F
..
:F:
..
|
.. | ..
:F: - Cl - :F:
.. | ..
|
..
:F:
..
🧪 Explanation
Chlorine pentafluoride (ClF₅) is an interhalogen compound where chlorine (Cl) is the central atom bonded to five fluorine (F) atoms. To draw its Lewis structure, we follow these steps:
1. Count Valence Electrons
- Chlorine (Cl) has 7 valence electrons.
- Each fluorine (F) also has 7 valence electrons.
- Total valence electrons = 7 (Cl) + 5 × 7 (F) = 42 electrons.
2. Determine the Central Atom
- Chlorine is less electronegative than fluorine, so it becomes the central atom.
3. Form Single Bonds
- Connect each of the five fluorine atoms to chlorine with a single bond.
- Each bond uses 2 electrons × 5 = 10 electrons.
4. Distribute Remaining Electrons
- Subtracting the bonding electrons: 42 − 10 = 32 electrons left.
- Each fluorine needs 6 more electrons to complete its octet (3 lone pairs).
- 5 fluorines × 6 electrons = 30 electrons used.
- 2 electrons remain, which are placed as a lone pair on chlorine.
5. Check Octet Rule and Expanded Octet
- Each fluorine has 8 electrons (1 bond + 3 lone pairs).
- Chlorine has 10 electrons around it (5 bonds + 1 lone pair), which is acceptable because it can expand its octet (period 3 element).
6. Molecular Geometry
- The electron geometry is octahedral (6 regions of electron density).
- The molecular shape is square pyramidal due to one lone pair on chlorine.
This structure satisfies all bonding and electron count rules, and it reflects the actual geometry and reactivity of ClF₅.
