The Correct Answer and Explanation is:
4.79
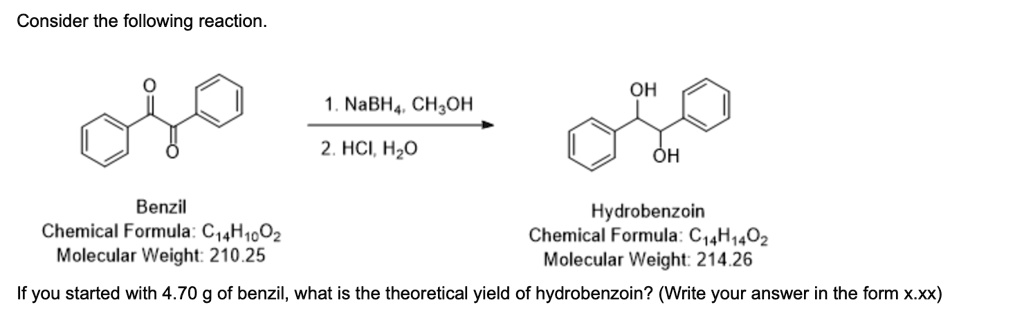
To determine the theoretical yield of hydrobenzoin, we first need to calculate the number of moles of the starting material, benzil. The theoretical yield represents the maximum possible mass of product that can be formed from the given amount of reactant, assuming the reaction proceeds to completion with 100% efficiency.
The calculation follows these steps:
- Calculate the moles of benzil:
We use the formula: Moles = Mass / Molecular Weight.
The given mass of benzil is 4.70 g.
The molecular weight of benzil is 210.25 g/mol .
Moles of benzil = 4.70 g / 210.25 g/mol ≈ 0.022354 moles. - Determine the moles of hydrobenzoin:
The chemical equation shows the conversion of one molecule of benzil to one molecule of hydrobenzoin. This indicates a stoichiometric ratio of 1:1 between the reactant and the product. Therefore, the number of moles of hydrobenzoin that can be theoretically produced is equal to the number of moles of benzil that we started with.
Moles of hydrobenzoin = Moles of benzil ≈ 0.022354 moles. - Calculate the theoretical mass of hydrobenzoin:
Now, we convert the moles of hydrobenzoin into mass (grams) using its molecular weight.
The formula is: Mass = Moles × Molecular Weight.
The molecular weight of hydrobenzoin is 214.26 g/mol .
Theoretical yield (mass) of hydrobenzoin = 0.022354 moles × 214.26 g/mol ≈ 4.789 g.
The question asks for the answer to be written in the form x.xx, which means we need to round our result to two decimal places. Rounding 4.789 g to two decimal places gives 4.79 g.
