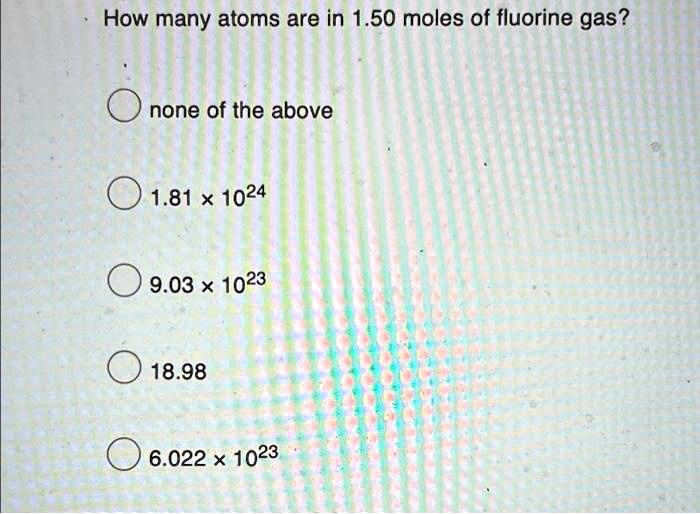
The Correct Answer and Explanation is:
The correct answer is 1.81 x 10^24.
To solve this problem, you need to use Avogadro’s number and understand the chemical nature of fluorine gas. Here is a step-by-step explanation of the calculation.
First, it is essential to know the chemical formula for fluorine gas. Fluorine is a halogen and one of the seven diatomic elements, which means it exists naturally as a molecule containing two atoms. Therefore, the chemical formula for fluorine gas is F₂, not simply F. This is a critical detail because the question asks for the total number of atoms, not the number of molecules.
The next step involves Avogadro’s number, which is the constant value of 6.022 x 10^23. This number represents the quantity of particles (such as atoms or molecules) in one mole of a substance.
The calculation can be broken down into two main parts:
- Calculate the number of F₂ molecules.
To find the number of molecules in 1.50 moles of fluorine gas, you multiply the number of moles by Avogadro’s number.
Number of molecules = (moles of substance) × (Avogadro’s number)
Number of F₂ molecules = 1.50 mol × (6.022 x 10^23 molecules/mol) = 9.033 x 10^23 molecules.
Note that this intermediate result, 9.03 x 10^23, is one of the answer choices. This is a common trap for students who forget that fluorine gas is diatomic and stop the calculation here. - Calculate the total number of fluorine atoms.
Since each molecule of fluorine gas (F₂) contains two fluorine atoms, you must multiply the number of molecules by two to find the total atom count.
Number of atoms = (Number of F₂ molecules) × (2 atoms per molecule)
Number of atoms = (9.033 x 10^23) × 2 = 18.066 x 10^23 atoms.
To present this value in proper scientific notation, we adjust the decimal point one place to the left and increase the exponent by one, which gives us 1.8066 x 10^24 atoms. The initial value given in the problem, 1.50 moles, has three significant figures, so the final answer should be rounded to three significant figures. Rounding 1.8066 x 10^24 gives us 1.81 x 10^24 atoms. This matches the correct option.
