A solution of H2SO4(aq) with a molal concentration of 3.97 m has a density of 1.216 g/mL.
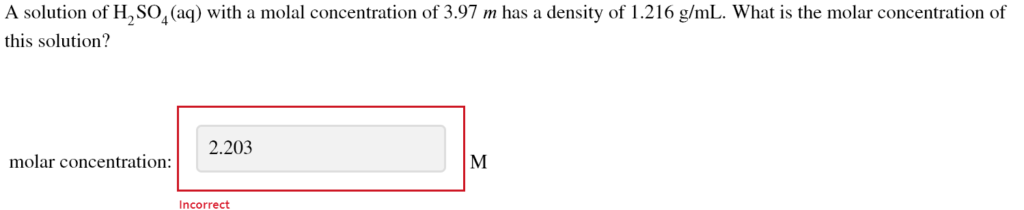
The Correct Answer and Explanation is:
3.47 M
To determine the molar concentration (molarity) from the molal concentration (molality), we need to find the number of moles of solute in one liter of the solution. The key difference between these two concentration units is their denominator: molarity uses the volume of the solution, while molality uses the mass of the solvent.
Here is the step by step conversion:
- Assume a Basis: To make the calculation straightforward, we will assume we have a sample of the solution that contains exactly 1 kg (1000 g) of solvent (water).
- Find Moles of Solute: The given molality is 3.97 m, which means there are 3.97 moles of H₂SO₄ for every 1 kg of water. So, our assumed sample contains 3.97 moles of H₂SO₄.
- Find Mass of Solute: We need to calculate the mass of 3.97 moles of H₂SO₄. First, we find the molar mass of H₂SO₄:
- H: 2 × 1.008 g/mol = 2.016 g/mol
- S: 1 × 32.06 g/mol = 32.06 g/mol
- O: 4 × 16.00 g/mol = 64.00 g/mol
- Molar Mass = 98.076 g/mol
Now, calculate the mass of the solute:
Mass of H₂SO₄ = 3.97 mol × 98.076 g/mol = 389.36 g
- Find Total Mass of Solution: The total mass of the solution is the sum of the mass of the solvent and the mass of the solute.
Total Mass = 1000 g (solvent) + 389.36 g (solute) = 1389.36 g - Find Volume of Solution: Using the given density of the solution (1.216 g/mL), we can find the volume.
Volume = Mass / Density = 1389.36 g / 1.216 g/mL = 1142.56 mL
To calculate molarity, this volume must be in liters:
Volume = 1142.56 mL / 1000 mL/L = 1.14256 L - Calculate Molarity: Molarity is moles of solute divided by liters of solution.
Molarity (M) = 3.97 mol / 1.14256 L = 3.474 M
Rounding to three significant figures, which is consistent with the given molality, the molar concentration is 3.47 M.
