COO
^+ H_3N-C-H \text{A.} CH_3 C
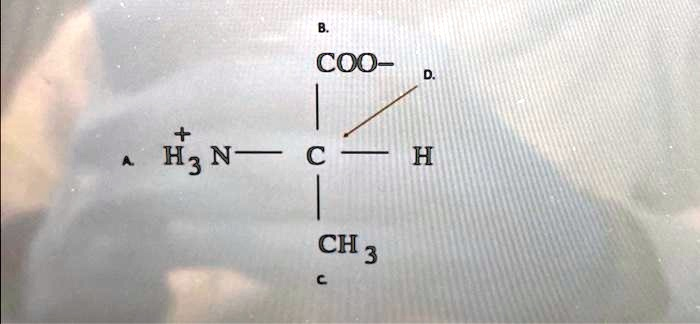
The Correct Answer and Explanation is:
Correct Answer: Alanine
Explanation:
The chemical structure shown in the image is the amino acid Alanine.
Amino acids are the fundamental building blocks of proteins. All 20 common amino acids share a core structure consisting of a central carbon atom, known as the alpha-carbon, bonded to four distinct groups:
- An amino group (-NH₂)
- A carboxyl group (-COOH)
- A hydrogen atom (-H)
- A variable side chain, or R-group, which is unique to each amino acid.
In the provided image, we can identify these components. The central carbon (C) is bonded to:
- An amino group, labeled A. It is shown in its protonated form (⁺H₃N-), carrying a positive charge.
- A carboxyl group, labeled B. It is depicted in its deprotonated, or carboxylate, form (COO⁻), carrying a negative charge.
- A hydrogen atom (H).
- A side chain (R-group), labeled C, which is a methyl group (-CH₃).
The identity of an amino acid is determined by its side chain. The amino acid with a methyl group as its R-group is Alanine. Because its side chain is composed only of carbon and hydrogen, Alanine is classified as a nonpolar, aliphatic amino acid. Its small, non-reactive side chain makes it chemically inert and versatile in protein structures.
The molecule is shown in its zwitterionic form. At physiological pH (around 7.4), the basic amino group is protonated (gains a proton, H⁺), and the acidic carboxyl group is deprotonated (loses a proton). This results in a molecule with both a positive and a negative charge but an overall neutral charge. This zwitterionic state is the predominant form of amino acids in neutral aqueous solutions.
Furthermore, the alpha-carbon in Alanine is a chiral center because it is attached to four different groups. This means Alanine can exist in two mirror-image forms (enantiomers), L-Alanine and D-Alanine, with L-Alanine being the form incorporated into proteins in living organisms.
