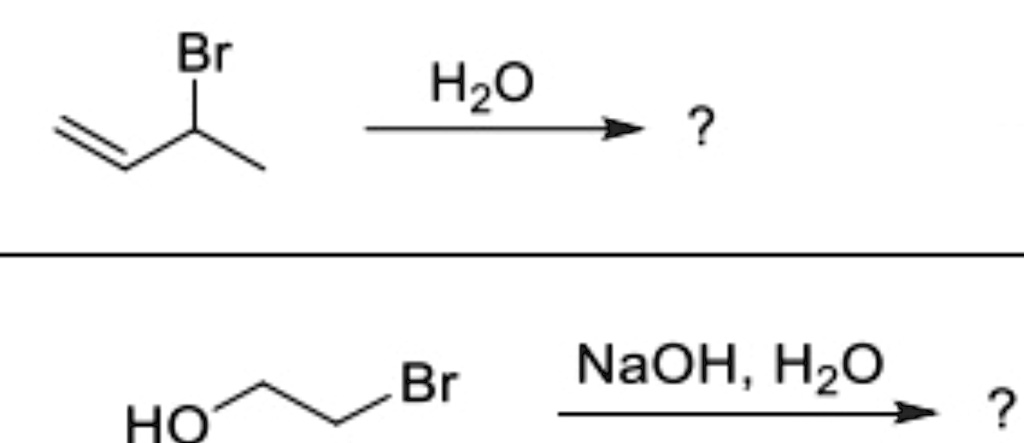
The Correct Answer and Explanation is:
It seems that the image you’ve provided shows two organic reactions involving a bromine (Br) atom attached to carbon atoms in a molecule. Let me break it down:
- First Reaction (top):
- The structure shown is an alkene (double bond between two carbon atoms) with a bromine atom attached to one of the carbons.
- The reagent is water (H2O).
- In this reaction, water will react with the alkene via hydrohalogenation. The double bond is broken, and water will add across the double bond, resulting in the formation of an alcohol.
- Product: The product would be an alcohol where one of the carbon atoms that was part of the original double bond now has a hydroxyl group (-OH) attached to it.
- Second Reaction (bottom):
- The starting material is a bromo-alcohol (HOCH2-CH2Br), with the hydroxyl group (-OH) on one carbon and the bromine atom on another.
- The reagents are sodium hydroxide (NaOH) and water (H2O).
- In this reaction, the hydroxide ion (OH-) from NaOH will undergo nucleophilic substitution on the carbon attached to the bromine (C-Br bond). The bromine atom will leave, and the hydroxide will take its place, forming an alcohol.
- Product: The product is an alcohol where the bromine is replaced by a hydroxyl group (-OH), resulting in a dihydroxy compound.
Both reactions involve substitution or addition mechanisms that lead to the formation of alcohols from the starting compounds.
