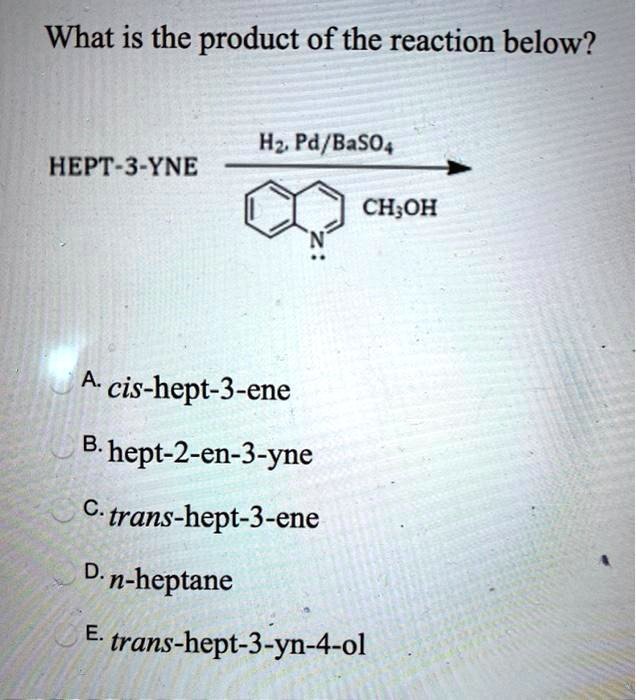
What is the product of the reaction below? H2 Pd/ BaSO4 HEPT-3-YNE CH3OH A. cis-hept-3-ene B. hept-2-en-3-yne C. trans-hept-3-ene D. n-heptane E. trans-hept-3-yn-4-ol
The Correct Answer and Explanation is:
The correct answer is A. cis-hept-3-ene.
This chemical transformation is a catalytic hydrogenation of an alkyne. The specific set of reagents used—H2 gas, palladium on barium sulfate (Pd/BaSO4), and quinoline (the nitrogen-containing aromatic compound shown)—is collectively known as Lindlar’s catalyst. This is a “poisoned” or deactivated catalyst system designed for a specific purpose.
The starting material is hept-3-yne, which is an internal alkyne with a seven-carbon chain and a triple bond between carbon 3 and carbon 4. The chemical structure is CH3-CH2-C≡C-CH2-CH2-CH3.
The role of Lindlar’s catalyst is to selectively reduce an alkyne to an alkene. The poison, quinoline, deactivates the palladium catalyst just enough to prevent the reaction from proceeding further to reduce the alkene to an alkane (n-heptane).
Crucially, the hydrogenation with Lindlar’s catalyst occurs with specific stereochemistry. The reaction mechanism involves the alkyne adsorbing onto the surface of the solid palladium catalyst. Then, two hydrogen atoms are delivered to the same face of the triple bond. This is known as a syn-addition.
When syn-addition occurs on an internal alkyne like hept-3-yne, the two substituents attached to the triple bond (in this case, an ethyl group and a propyl group) end up on the same side of the newly formed double bond. This geometric arrangement defines a cis or (Z) isomer. Therefore, the reduction of hept-3-yne with Lindlar’s catalyst yields cis-hept-3-ene.
The other options are incorrect. Trans-hept-3-ene would be formed by an anti-addition reaction, such as using sodium metal in liquid ammonia. N-heptane would be the product of complete hydrogenation using a more active catalyst like palladium on carbon (Pd/C). The remaining options involve incorrect functional groups or bond arrangements not produced by this reaction.
