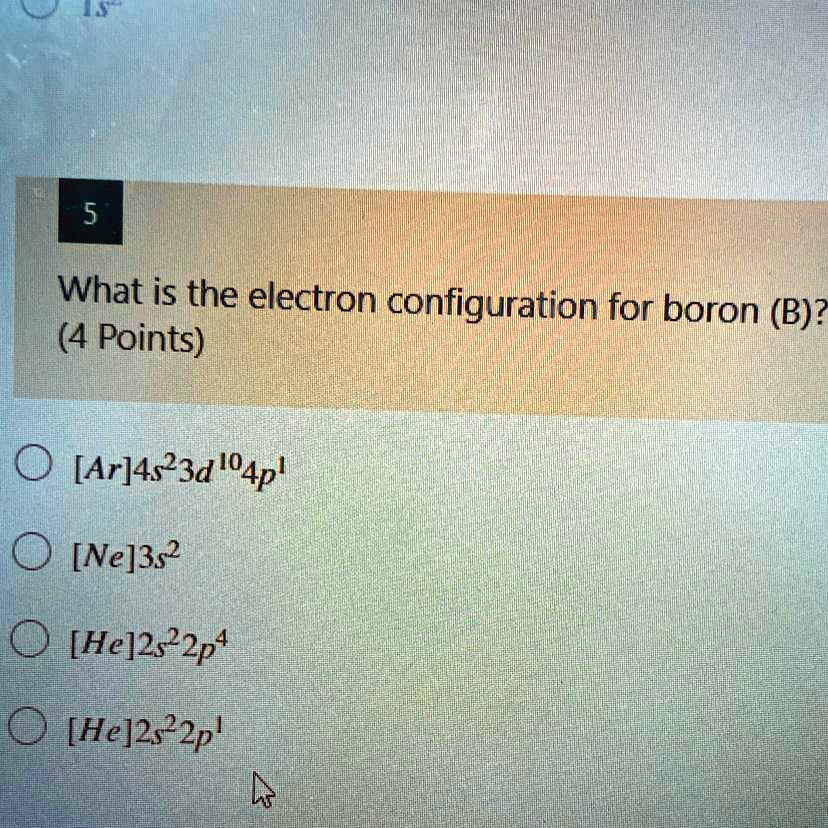
What is the electron configuration for boron (B)? (4 Points) [Ar]4s²3d¹?4p¹ [Ne]3s² [He]2s²2p? [He]2s²2p¹
The Correct Answer and Explanation is:
The correct electron configuration for boron (B) is [He]2s²2p¹.
To determine the electron configuration of an element, we must first identify its atomic number from the periodic table. The atomic number represents the number of protons in an atom’s nucleus and, for a neutral atom, also equals the number of electrons. Boron (B) has an atomic number of 5, which means a neutral boron atom contains 5 electrons.
Electron configuration describes how these electrons are distributed among the various atomic orbitals. We fill these orbitals according to the Aufbau principle, which states that electrons occupy the lowest energy orbitals available first. The order of increasing energy for the first few orbitals is 1s, 2s, 2p, 3s, and so on.
Let’s place Boron’s 5 electrons into these orbitals:
- The first two electrons fill the lowest energy orbital, the 1s orbital. This gives us 1s².
- The next two electrons fill the next available orbital, the 2s orbital. This gives us 2s².
- The final, fifth electron enters the next available orbital, which is the 2p orbital. This gives us 2p¹.
Combining these parts gives the full electron configuration for boron: 1s²2s²2p¹.
The question provides answers in noble gas shorthand notation. This method simplifies the configuration by using the symbol of the preceding noble gas to represent the core electrons. The noble gas that comes before boron is Helium (He), which has the electron configuration 1s². We can therefore replace the “1s²” portion of boron’s configuration with “[He]”. The remaining electrons, which are the valence electrons, are written out. This results in the shorthand configuration [He]2s²2p¹. This matches the last option provided. The other options are incorrect as they represent different elements: [Ne]3s² is magnesium (12 electrons), and [He]2s²2p⁴ is oxygen (8 electrons).
