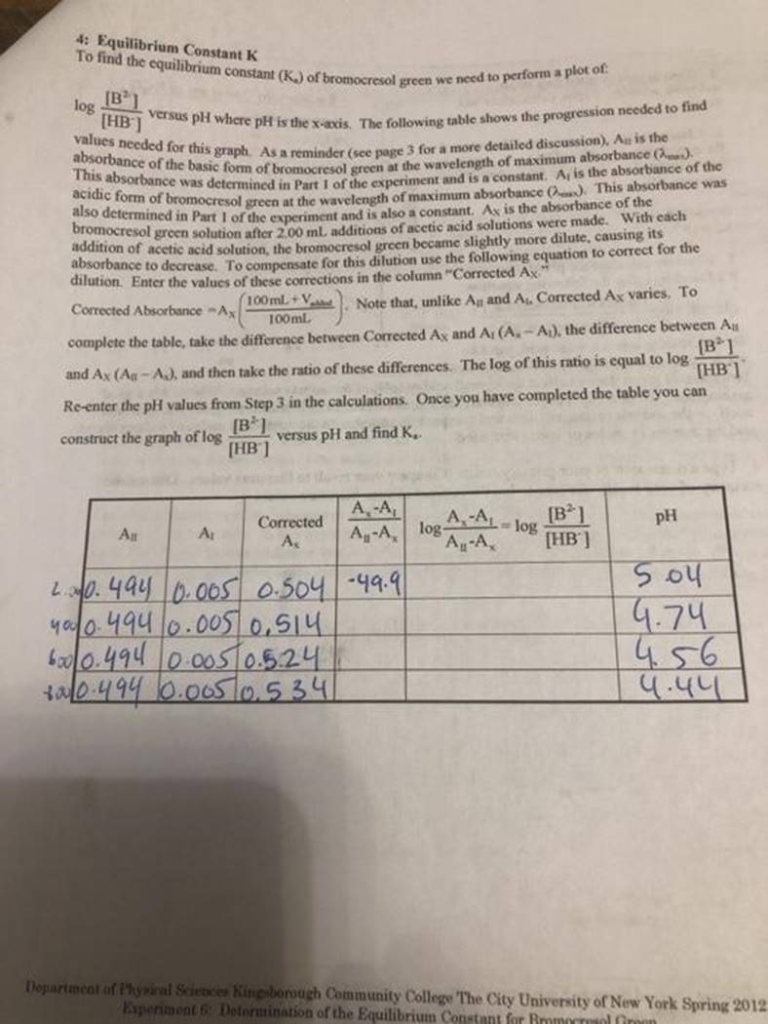
Equilibrium Constant K To find the equilibrium constant (K) of bromocresol green we need to perform a plot of [B] log [HB] versus pH where pH is the x-axis. The following table shows the progression needed to find absorbance of the basic form of bromocresol green at the wavelength of maximum absorbance). values needed for this graph. As a reminder (sce pape 3 for a more detailed discussion). A is the This absorbance was determined in Part 1 of the experiment and is a constant. Aris the absorbance of the acidic form of bromocresol green at the wavelength of maximum absorbance (2) This absorbance was 100ml bromocresol green solution after 2.100 mL dditions of acetic acid solutions were made. With each addition of acetic acid solution, the bromocresol green became slightly more dilute, causing its absorbance to decrease. To compensate for this dilution use the following equation to correct for the dilution Enter the values of these corrections in the column Corrected Ax” 100 ml. + Corrected Absorbance-AX Note that, unlike Aland A. Corrected Ax varies. To complete the table, take the difference between Corrected Ax and A, (A.-A), the difference between A [B1 and Ax (A-AJ. and then take the ratio of these differences. The log of this ratio is equal to log THB) Re-enter the pH values from Step 3 in the calculations. Once you have completed the table you can [BPI construct the graph of log [HB] versus pH and find K. A-A Corrected pH All A, [B] log [HB] ?. A-A log A-A “A-A, 2.0.494 0.005 0.504 -49.9 40000 4940.005 0.514 600/0.494 0.005 0.5.24 *0.494 0.005 0. 534 Sou 9.74 ? 4.56 4.44 ?? Department of Physical Science Kinerborough Community College ‘The City University of New York Spring 2012 Experiment 6: Determination of the Equilibrium Constant for more in
The Correct Answer and Explanation is :
To determine the equilibrium constant (K) for bromocresol green (BCG), you’ll follow a series of steps and calculations that involve absorbance data, pH values, and the dilution of the bromocresol green solution. The equilibrium constant (K) is related to the concentrations of the protonated (HB) and unprotonated (B) forms of bromocresol green, and the goal of this experiment is to plot the log of the ratio of these concentrations versus the pH.
Step-by-step Breakdown:
- Absorbance Data:
- Absorbance values for both the acidic form (HB) and basic form (B) are measured at the wavelength of maximum absorbance.
- Absorbance (A) will decrease as the solution becomes more dilute with the addition of acetic acid, which shifts the equilibrium toward more protonated forms (HB).
- You are provided with an equation to correct for dilution effects:
[
A_{\text{corrected}} = A \times \left( \frac{100\, \text{mL}}{100 + \text{mL of acid added}} \right)
] - This correction accounts for the decreasing concentration of bromocresol green as acetic acid is added.
- Calculate Differences:
- Calculate the difference in absorbance between the corrected absorbance (A_corrected) and the absorbance of the basic form (A_B):
[
\Delta A = A_{\text{corrected}} – A_B
] - The value of A_B is constant for each pH value and represents the absorbance of the unprotonated form of bromocresol green.
- Concentration Ratio:
- Once the difference between the corrected absorbance and the basic form absorbance is calculated, the ratio of the concentrations of HB and B can be determined using the absorbance values. This ratio is related to the pH:
[
\frac{[HB]}{[B]} = \frac{A_{\text{corrected}} – A_B}{A_A – A_B}
] - Taking the logarithm of this ratio gives log [HB].
- Plot and Calculation:
- Plot the log of [HB] versus the pH values from the table.
- The equilibrium constant (K) can be derived from the slope of the line in the plot.
The graph you construct will give a straight line whose slope is directly related to the equilibrium constant (K) through the following equation:
[
\log K = \text{slope}
]
This relationship arises from the fact that the equilibrium constant for a weak acid, like bromocresol green, is related to the pH and the ratio of the concentrations of the protonated and unprotonated forms. From the slope, K can be calculated, giving you the equilibrium constant for bromocresol green under the conditions of the experiment.