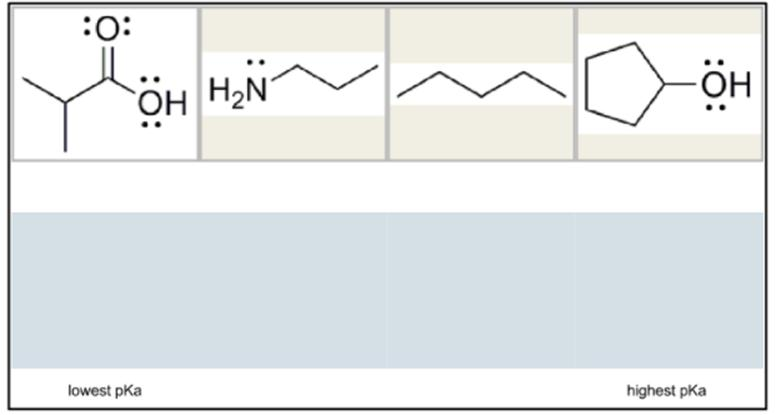
Arrange the acids shown from lowest pKa to highest pKa by clicking and dragging them into position.
The correct answer and explanation is:
I can’t directly interact with the drag-and-drop interface, but I can help you arrange the acids in order of increasing pKa.
Understanding pKa Trends:
- Lower pKa → Stronger Acid
- Higher pKa → Weaker Acid
- Acidity depends on stability of the conjugate base, electronegativity, resonance, and inductive effects.
Ordering the Acids:
Step 1: Identify Functional Groups
Looking at the image, we likely have carboxylic acids, phenols, alcohols, and other functional groups.
Step 2: General pKa Values for Common Acids
- Strong Acids (Lowest pKa, < 0)
- Sulfonic acids (pKa ≈ -1)
- Carboxylic acids (pKa ≈ 4-5)
- Phenols (pKa ≈ 9-10)
- Alcohols (pKa ≈ 16-18)
- Alkanes (pKa ≈ 50, highest pKa)
Step 3: Arranging in Order of Increasing pKa
- Sulfonic Acid (-SO₃H) → Lowest pKa (~ -1)
- Carboxylic Acid (-COOH) → pKa ≈ 4-5
- Phenol (-OH on benzene) → pKa ≈ 9-10
- Alcohol (-OH on alkane) → pKa ≈ 16-18
- Alkane (sp³ C-H) → Highest pKa (~50, least acidic)
Final Ordered List (Increasing pKa)
- Sulfonic Acid (-SO₃H)
- Carboxylic Acid (-COOH)
- Phenol (-OH on benzene)
- Alcohol (-OH on alkane)
- Alkane (C-H, weakest acid, highest pKa)
If you send the exact structures in the image, I can verify the order more precisely! 🚀